Neuralace Medical’s Axon Therapy gets FDA clearance, revolutionising PDN treatment

The therapy’s efficacy has been validified via a comprehensive RCT. Credit: ream sukanya via Shutterstock. Neuralace Medical, a pioneering company in the medical technology sector, has achieved a significant milestone with the recent US Food and Drug Administration (FDA) clearance of its innovative Axon Therapy. This breakthrough is specifically targeted at treating chronic painful diabetic […]
New algorithm by PulseAI to improve ECG diagnostics

PulseAI’s innovative algorithm promises to enhance diagnostic accuracy. Credit: SFAM_Photo via Shutterstock. PulseAI has unveiled a new artificial intelligence (AI) algorithm poised to redefine the landscape of cardiac health monitoring. The new technology combines AI with electrocardiogram (ECG) data, empowering individuals to proactively assess and understand their heart health with unprecedented precision. PulseAI’s innovative algorithm […]
Avoid “narrow national interest” in fighting next pandemic, WHO chief says

Dr. Tedros Adhanom Ghebreyesus was speaking at a panel discussing how healthcare systems should prepare for “Disease X”, at the ongoing World Economic Forum (WEF) being held in Davos. Image credit: GettyImages/Bloomberg. “Narrow national interest should not come in the way” of fighting against the next big public health threat, says World Health Organisation (WHO) […]
Darmiyan’s brain test, Brainsee, has gained FDA approval

6,500,000 patients ages 65 years and older are currently living with Alzheimer’s disease in the US. Credit: STEKLO via Shutterstock. January is Alzheimer’s Awareness Month, as designated by the Alzheimer Society of Canada. The society’s objective is to prompt individuals and organisations to educate themselves on dementia, including its risk factors, prevention and treatment. Although […]
Positive trial results for Orlucent’s handheld melanoma detection device
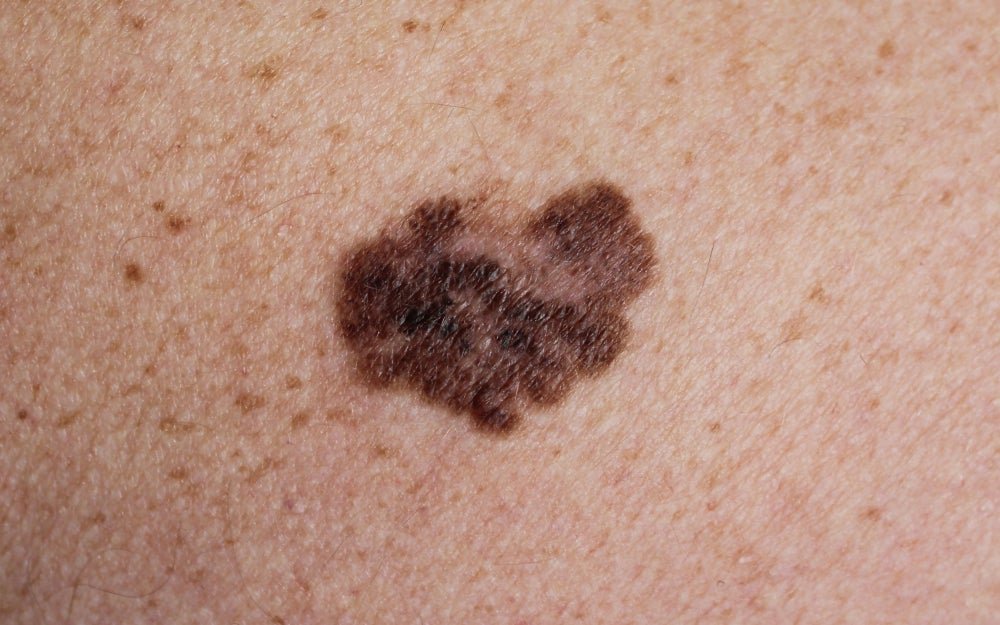
Melanoma is the third most common type of skin cancer, according to the US Centers for Disease Control and Prevention. Image credit: Shutterstock/Nasekomoe. Orlucent has reported positive results from a study evaluating its handheld point-of-care molecular skin fluorescence imaging (mSFI) system for detecting a type of skin cancer. Data from the first-in-human studywas published in Pigment […]
Clarius wins CE mark for wireless handheld ultrasound
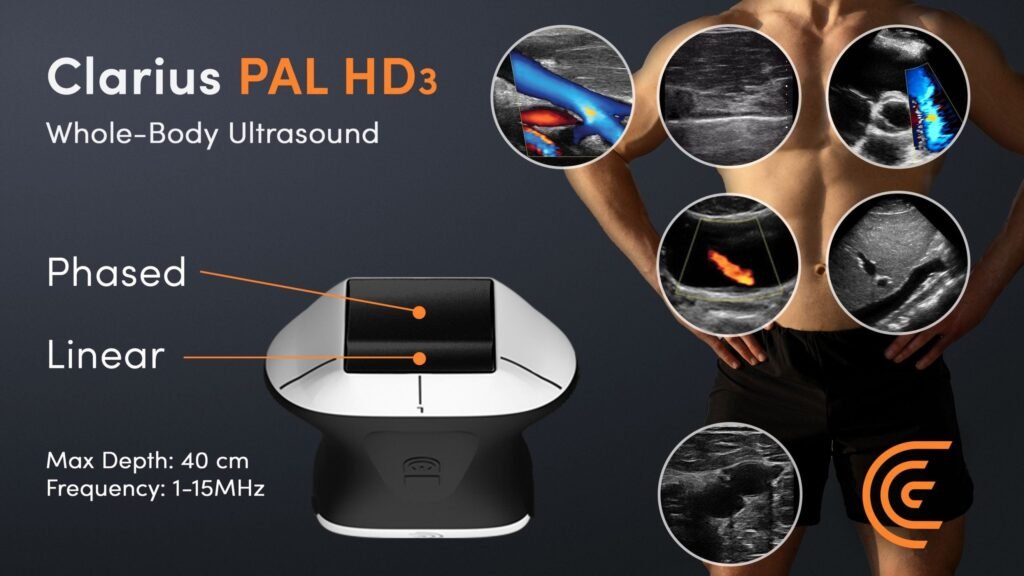
The Claruis’ handheld ultrasound device can provide high-resolution images up to 40 cm in depth from the skin surface. Image Credit: Clarius Mobile Health. Canadian Clarius Mobile Health has received a CE mark for its wireless handheld whole-body ultrasound scanner, Clarius PAL HD3. Concurrently the company has also launched the device in the European Union […]
Sensydia completes study for Cardiac Assessment Platform
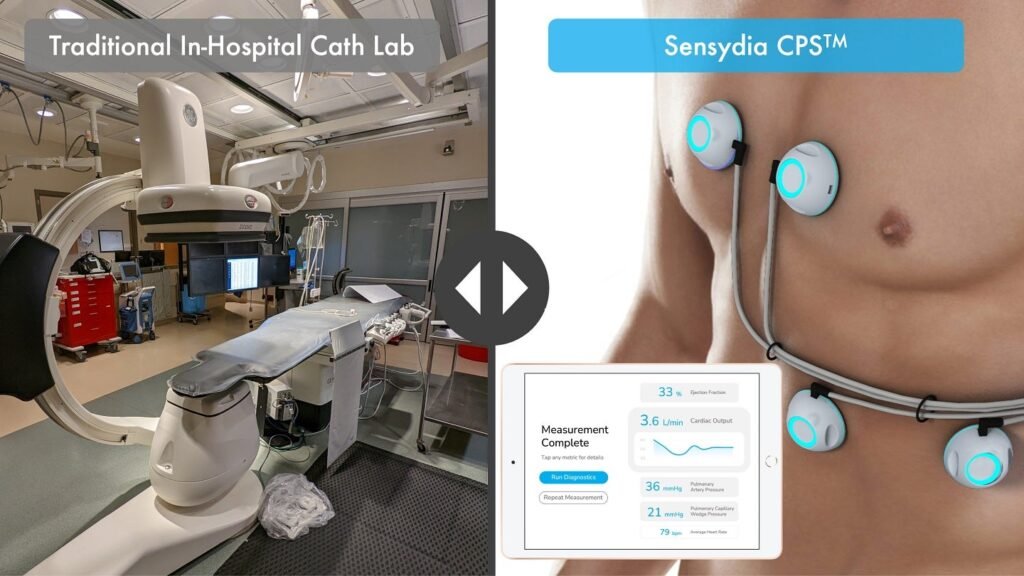
Sensydia’s non-invasive pulmonary pressure measurement. Credit: Business Wire/Sensydia. Sensydia has announced the completion of a pivotal study of the Cardiac Performance System (CPS) involving 50 subjects, marking a significant step towards the introduction of a non-invasive device for measuring pulmonary pressure. The study, conducted at the University of Minnesota, gathered data for the system, which […]
IMVARIA bags FDA authorisation for AI lung fibrosis diagnostic

GlobalData has identified idiopathic lung fibrosis (ILF) as an area of unmet need. Credit: ALPA PROD via Shutterstock. The US Food and Drug Administration (FDA) has given the green light to US-based IMVARIA for its lead product Fibresolve, an artificial intelligence (AI)-based lung fibrosis biomarker. The AI-driven tool helps to diagnose lung fibrosis, particularly idiopathic […]
Apple redesigns iWatch responding to federal ban of pulse oximetry feature

A letter filed to the US Court of Appeals by Masimo confirms that Apple has redesigned its iWatch features to fall outside of the scope of Masimo’s patents. Credit: Shuttertstock/agencies US tech giant Apple is set to pull a feature from its brand of smartwatches that enables measurement of a user’s blood oxygen levels, following […]
Noctrix Health secures funding to advance Nidra TOMAC therapy

The Nidra TOMAC therapy received De Novo authorisation in April 2023. Credit: PRNewswire/Noctrix Health. Noctrix Health has secured $40m in a Series C financing round to commercialise its new Nidra Tonic Motor Activation (TOMAC) therapy for restless legs syndrome (RLS) in the US market. The funding round was led by Sectoral Asset Management and included […]
