National Health Service launches new ‘blood matching’ genetic test
The new test is expected to benefit nearly 18,000 eligible individuals. Credit: Robert DeLaRosa from Pixabay. The National Health Service (NHS) in the UK has introduced a ‘blood matching’ genetic test, marking a global first in personalised healthcare for patients with sickle cell disorder and thalassaemia. This new blood group genotyping test is expected to […]
Modular Medical submits MODD1 insulin pump for FDA clearance

The development marks a significant step for the company in launching its next-generation insulin pump. Credit: Towfiqu barbhuiya on Unsplash. Insulin delivery technology company Modular Medical has announced the premarket submission of its MODD1 insulin pump to the US Food and Drug Administration (FDA) to obtain 510(k) clearance. This marks a significant step for the […]
Dexcom begins work on new manufacturing facility in Ireland

The new facility aims to reduce costs and enhance sustainability by cutting delivery times. Credit: Diabetesmagazijn.nl on Unsplash. rtCGM systems provider DexCom has commenced construction on its first manufacturing facility in Europe, located in Athenry, County Galway, Ireland. Expected to generate more than 1,000 jobs, including across human resources (HR), finance and engineering disciplines, the […]
Summa Therapeutics performs first injectable angioplasty in PAD patients
The number of angioplasty procedures in the United States is forecast to reach 2,103,798 in 2030. Credit: MAD.vertise via Shutterstock. US-based device company Summa Therapeutics has performed the first injectable angioplasty with its Finesse InjectableTM balloon catheter on patients with peripheral arterial disease (PAD). The design of the catheter aims to increase the efficiency […]
OrganOx and eGenesis advance in porcine liver replacement trial

The eGenesis study leans on another pre-clinical study, published in the journal Nature that was similarly able to show that porcine livers in certain circumstances could be used in place of human organs. Credit: Shutterstock / Shidlovski OrganOx and eGenesis have announced the successful completion of an extracorporeal perfusion of a brain-dead research donor using […]
Nectero starts clinical trial for endovascular aneurysm stabilisation system
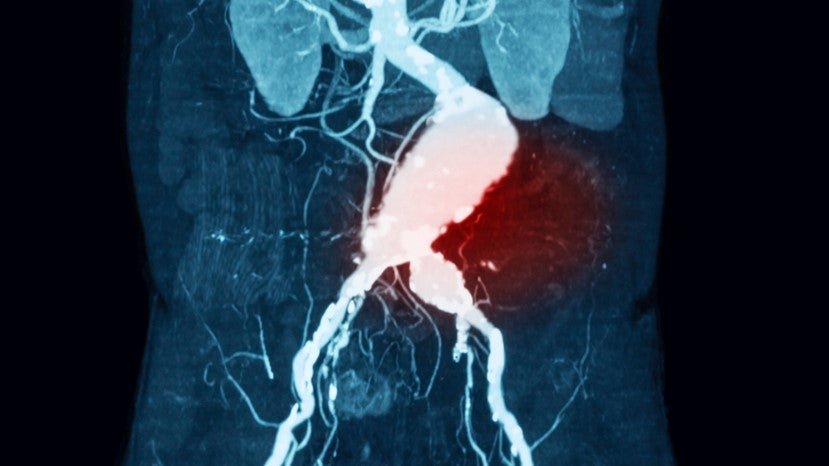
Nectero EAST device received a fast-track designation from the US Food and Drug Administration (FDA) in August 2023. Image Credit: Suttha Burawonk / Shutterstock. Arizona-based Nectro Medical has initiated a Phase II/III trial evaluating the safety and efficacy of its endovascular aneurysm stabilisation treatment (Nectero EAST). Nectero EAST is a single-use, endovascular system made up […]
Kallisio’s 3D printed oral stent gains US FDA clearance for cancer
The 3D stent can ensure radiation delivery, particularly in intensity-modulated radiation therapy. Credit: Elnur/shutterstock.com. Kallisio has received US Food and Drug Administration (FDA) 510(k) clearance for its 3D-printed oral stent solution, Stentra, designed for precise radiation therapy for head and neck cancer patients. The device aims to protect healthy tissue by immobilising organs at risk […]
Tempus launches MRD assay for colorectal cancer research
The new assay incorporates algorithms to filter out artifacts, clonal hematopoiesis of indeterminate potential (CHIP) and germline variants. Credit: CDC on Unsplash. Technology company Tempus has announced the launch of the new minimal residual disease (MRD) assay, xM, designed for colorectal cancer (CRC) research. The xM assay is a tumour-naïve, plasma-based test that detects circulating […]
Calcivis to introduce preventive dental technology in US
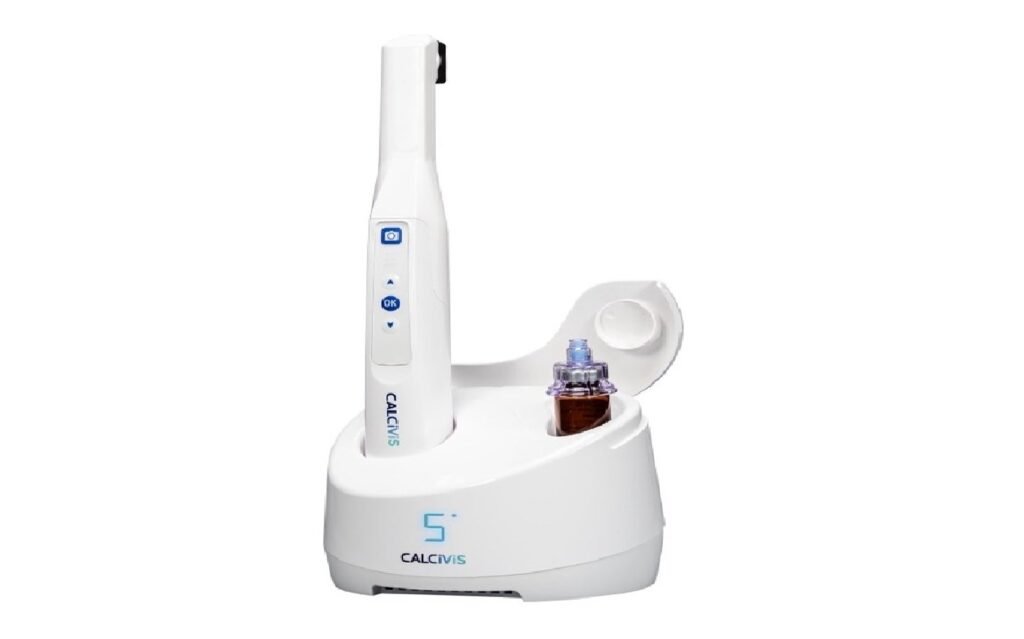
Calcivis’ bioluminescent dental imaging system. Credit: Business Wire/Calcivis. Medical device company Calcivis is set to launch its new preventive bioluminescent dental imaging system in the US. The move comes after the company secured final-stage US Food and Drug Administration (FDA) pre-market approval (PMA) for its new ergonomic, wireless, handheld imaging device, paving the way for […]
FDA approves expanded MRI labelling for Abbott’s neurostimulator
The development allows patients implanted with the device to undergo full-body Magnetic resonance imaging (MRI) scans. Credit: Michal Jarmoluk from Pixabay. The US Food and Drug Administration (FDA) has granted approval of expanded MRI labelling for Abbott’s Proclaim DRG neurostimulation system. This development allows patients implanted with the device to undergo full-body MRI scans. The […]
