Delfi launches new assay to monitor cancer treatment

The test is designed for the non-invasive measurement of tumour burden. Credit: eczserapyilmaz/Shutterstock.com. US-based blood-based cancer detection test developer Delfi Diagnostics has introduced the new DELFI-Tumor Fraction (DELFI-TF) assay. DELFI-TF is a fragmentome-based, research-use-only (RUO) test designed for the non-invasive measurement of tumour burden. This assay is expected to play a crucial role in assessing […]
Synchron acquires stake in components maker Acquandas

Synchron’s Stentrode endovascular electrode array. Credit: Synchron/Business Wire. Brain-computer interface (BCI) technology company Synchron has acquired an equity stake in Acquandas, a specialist in high-precision components, for an undisclosed amount. The stake acquisition is expected to bolster Synchron’s position in the implantable medical device industry by integrating Acquandas’ advanced shape-memory, flexible metallic components into its […]
Excellence Awards Report 2023 – Medical Device Network

Credit: (LALAKA)/Shutterstock The Excellence Awards celebrate the greatest achievements and innovations in the industry. The programme provides a platform to recognize the people and companies that are driving change. Download the full report below The Awards Report includes: Has your company won an Excellence Award? Search to find out if your company is a winner […]
Diagnostic imaging device innovation landscape for 2024
This year is set to see increased demand for early disease diagnosis and personalised medicine. Credit: Terelyuk via Shutterstock. Advancements in software, hardware, and consumables in diagnostic imaging are being driven by the demand for early disease diagnosis and personalised medicine. According to insights from GlobalData, a leading data and analytics company, developments in AI, […]
Neuronoff implants injectable electrode in first-in-human study
A report by GlobalData estimates the global neuromodulation device market will be worth $11.4bn by 2033. Image credit: Shutterstock/adike. Neuronoff has completed the first implants of its Injectrode device in a first-in-human study evaluating its use in chronic pain management. The minimally invasive injectable electrode works by a type of neuromodulation called transcutaneous electrical nerve […]
Excellence Awards Report 2023 – Medical Device Network

Credit: (LALAKA)/Shutterstock The Excellence Awards celebrate the greatest achievements and innovations in the industry. The programme provides a platform to recognize the people and companies that are driving change. Download the full report below The Awards Report includes: Has your company won an Excellence Award? Search to find out if your company is a winner […]
Roche wins CE mark for PCOS diagnostic immunoassay

Roche’s Elecsys AMH blood test has been approved as a fertility marker to assess ovarian reserve levels. Image Credit: JHVEPhoto / Shutterstock. Roche Diagnostics has received a CE mark for Elecsys anti-müllerian hormone (AMH) plus immunoassay as a diagnostic blood test for PCOS. AMH is produced by granulosa cells in ovarian follicles. In patients with […]
Synergy raises $30m to advance artificial cervical disc

Symptomatic degenerative disc disease can cause chronic back or neck pain, and decreased mobility. Credit: staras via Shutterstock. Synergy Spine Solutions has closed a Series A funding round worth $30m, which will be used to advance its artificial cervical disc Synergy Disc through clinical trials and regulatory clearings. The funding, spearheaded by Amzak Health alongside […]
Philips TEE transducer secures 510(k) clearance from FDA
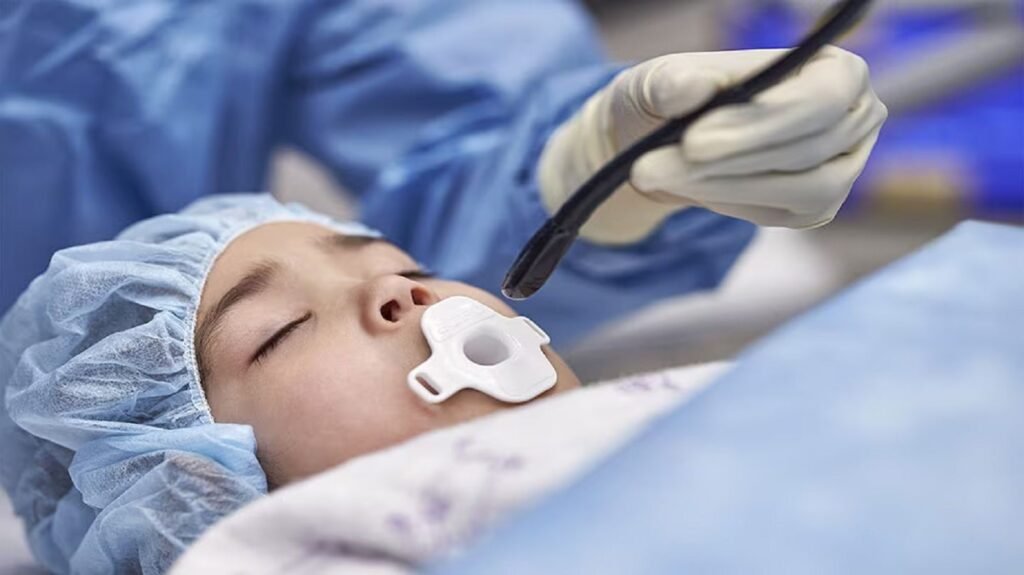
A patient undergoing diagnosis with X11-4t Mini 3D TEE transducer. Credit: Koninklijke Philips N.V. Philips has secured 510(k) clearance from the US Food and Drug Administration (FDA) for its latest transoesophageal echocardiography (TEE) transducer, designed to help improve cardiac care for more patients. The Philips X11-4t Mini 3D TEE transducer, which is 35% smaller than […]
UK MHRA expands medical device certification capacity

The latest development enhances the capacity of UK MHRA for ensuring device safety and performance. Credit: SEVENNINE_79 / Shutterstock.com. The UK Medicines and Healthcare products Regulatory Agency (MHRA) has expanded the country’s capacity to certify the safety and performance of medical devices. Two new UK approved bodies, LNE-GMED UK and Scarlet NB UK, have been […]
