FDA green lights StimLabs umbilical cord-derived wound graft
The wound care management market is forecast to generate $3.1bn in 2030 in the US. Credit: antoniodiaz via Shutterstock. The US Food and Drug Administration (FDA) has granted 510(k) clearance to US-based StimLabs wound care device Corplex P. The device claims to be the first graft derived from the human umbilical cord extracellular matrix (ECM), […]
Viz.ai’s aneurysm diagnostic tool is 97.2% predictive in ruling out stroke

The data from the abstracts demonstrate the use of Viz’s AI platform in improving workflows and disease detection. Image Credit: Golden Dayz / Shutterstock. US-based Viz.ai has released new trial data that assessed its artificial intelligence (AI) powered neurovascular technology to detect and diagnose neurology pathologies including acute ischemic stroke, cryptogenic stroke, unruptured incidental aneurysm […]
Delve Health and AliveCor team up on cardiac care
The KardiaMobile 6L allows patients and healthcare professionals to monitor heart rhythms and detect abnormalities. Credit: Golden Dayz/shutterstock.com. Delve Health has announced a collaboration to integrate AliveCor’s KardiaMobile 6L personal EKG device into its LiveCare Link + smart RPM gateway to improve cardiac care. This partnership aims to enhance research, expedite drug development processes and […]
BlueWind launches Revi implantable device in Ohio, US

The Revi device intends to mitigate the potential adverse events associated with other surgical UUI therapies. Credit: ARMMY PICCA/shutterstock.com. BlueWind Medical has announced the availability of its Revi implantable Tibial NeuroModulation (iTNM) device for urge urinary incontinence (UUI) in Springfield, Ohio, US. The patient-centric, minimally invasive solution is now offered at Ohio Valley Surgical Hospital. […]
AstraZeneca selects SOPHiA to expand HRD testing in Spain
SOPHiA GENETICS and AstraZeneca Spain are expanding HRD testing for ovarian cancer across Spain. Credit: Tatiana from Pixabay. AstraZeneca Spain has chosen cloud-native software company Sophia Genetics as its preferred partner for expanding HRD testing for ovarian cancer across Spain. SOPHiA GENETICS said that the programme, which began in 2022, initially enabled five laboratories in […]
Innova announces Laguna system’s early commercial use
The devices are intended for application in the peripheral vasculature. Credit: Peter Porrini/ Shutterstock.com. Innova Vascular has reported the successful early commercial use of its Laguna Thrombectomy System in patients. The Laguna Thrombectomy System comprises two devices and is designed to remove blood clots from patients’ vessels. It includes the Malibu Aspiration Catheter and the […]
Insulet gains CE mark for Omnipod 5’s combo with Abbott FreeStyle Libre sensor
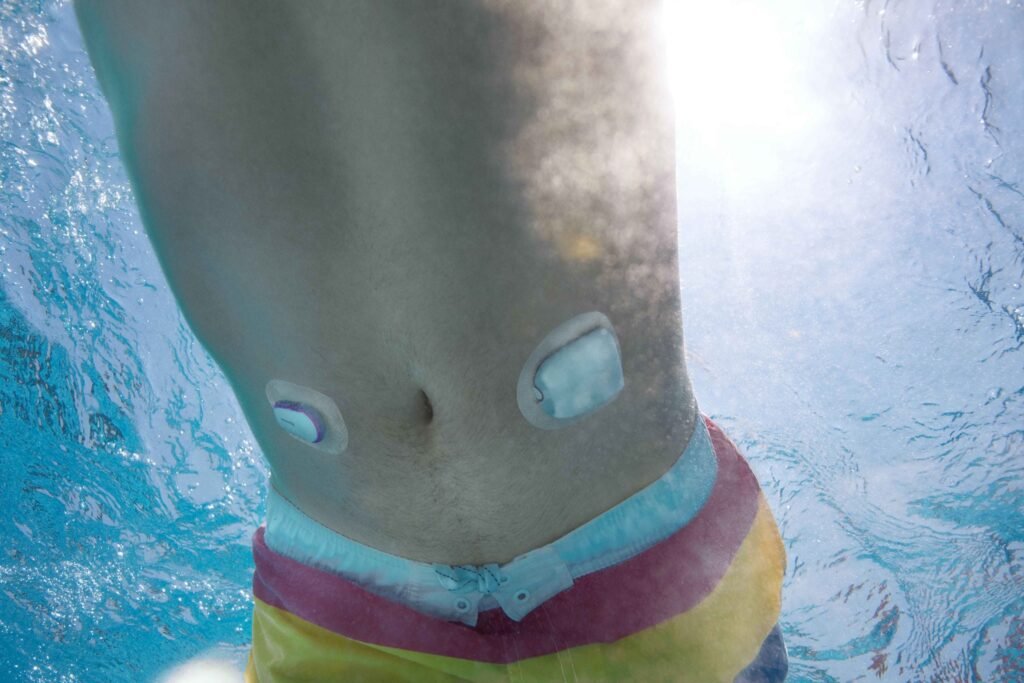
The Omnipod 5 can be used by adults and children aged two and older with type 1 diabetes. Image credit: Insulet. Patients in Europe who use the FreeStyle Libre 2 Plus sensor can now team it with Insulet’s Omnipod 5 after the latter device received CE mark approval for compatibility with Abbott’s continuous glucose monitor […]
Biocomposites initiates two-Phase II trials of STIMULAN VG in US
Diabetic foot osteomyelitis is a soft tissue infection that spreads into the bone. Credit: Alona Siniehina via Shutterstock. UK-based medical device company Biocomposites has announced the initiation of two Phase II clinical trials of its antibiotic carrier STIMULAN VG in the US. STIMULAN is a pharmaceutical-grade calcium sulphate biodegradable and absorbable bone graft substitute implant. […]
Tyber Medical’s anatomical plating system receives approval
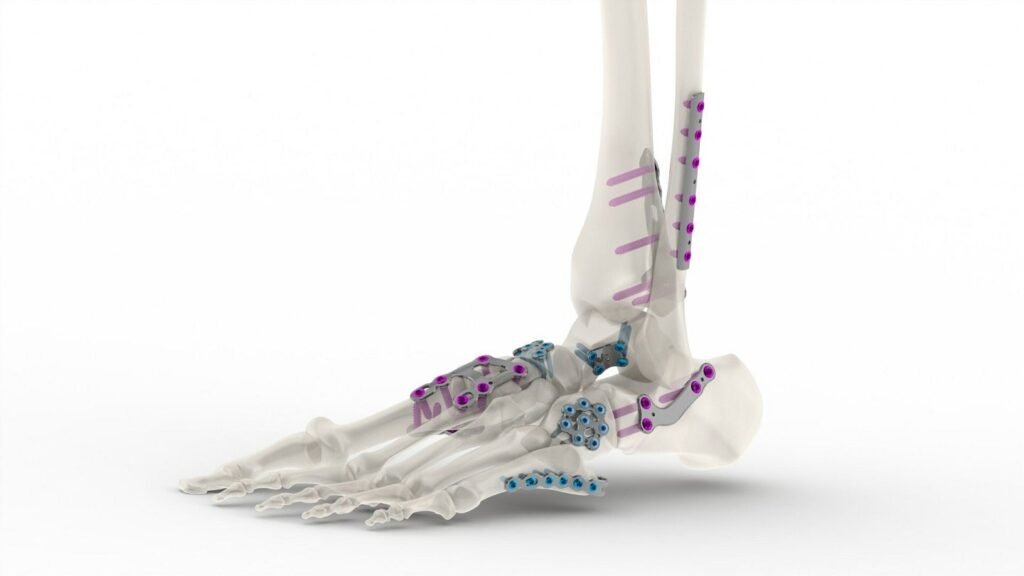
Tyber Medical’s anatomical plating system. Credit: Tyber Medical/ PRNewswire. Private-label orthopaedic implant provider Tyber Medical has received clearance from Health Canada for its anatomical plating system. The system previously received approval by the US Food and Drug Administration (FDA). Tyber Medical CEO and president Jeff Tyber said the company secured approval for a comprehensive plating […]
Zynex’s new NMES device secures FDA clearance
The new device enables the treatment of patients in a clinical or home setting. Credit: Gorodenkoff /Shutterstock.com. The US Food and Drug Administration (FDA) has approved medical technology company Zynex’s next-generation neuromuscular electrical stimulation (NMES) device, the M-Wave. This device is intended to aid in the recovery from surgery, manage chronic conditions, and enhance exercise […]
