Onward’s brain-computer interface tech joins FDA’s new TAP programme

Shares in the Euronext Brussels-listed company have risen sharply since the turn of the year following clinical and developmental milestones. Image credit: Shutterstock/thisdesign. After winning breakthrough device designation, Onward Medical has been accepted into the US Food and Drug Administration’s (FDA) total product life cycle advisory programme (TAP) for its brain-computer interface (BCI) technology. The […]
Enzymatica wins CE-certification for nasal spray ColdZyme

Enzymatica’s mouth spray ColdZyme has received CE-certification under the new European Union medical device regulation (MDR). Credit: stoatphoto via Shutterstock. Enzymatica’s mouth spray ColdZyme has secured CE-certification of Class III from Eurofins, a European-approved notified body for medical devices. Used to treat cold and flu-like infections, ColdZyme provides a barrier that protects the oral cavity […]
Philips and SyntheticMR launch AI-based quantitative brain imaging system

GlobalData projects AI in the diagnostic imaging market to reach $1.2bn by 2027. Credit: Jaray Sansed via Shutterstock. In the rapidly evolving field of medical diagnostics, Philips and Synthetic MR have embarked on a collaboration to enhance the diagnosis of neurological disorders using advanced quantitative brain imaging tools. This partnership aims to revolutionise the detection […]
Surgalign Holdings Inc Patent: Photocurable Liquid Adhesive Kit
According to GlobalData’s company profile on Surgalign, Prosthetic cardiac valves was a key innovation area identified from patents. Surgalign‘s grant share as of January 2024 was 58%. Grant share is based on the ratio of number of grants to total number of patents. Photocurable liquid adhesive kit with two containers Source: United States Patent and […]
PulseCath lands CE mark for mechanical circulatory support device
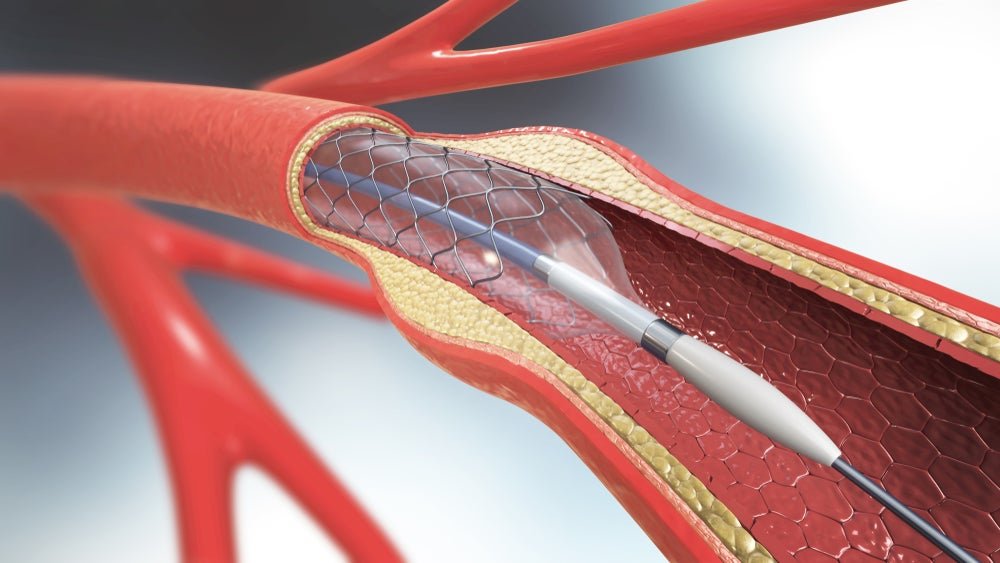
PulseCath’s iVAC 2L system is a percutaneous mechanical circulatory support (MCS) device. Image Credit: Christoph Burgstedt / Shutterstock. Netherlands-based PulseCath has received a CE mark under the EU Medical Device Regulation (MDR) for its mechanical circulatory support device, iVAC 2L. PulseCath’s iVAC 2L system is a percutaneous mechanical circulatory support (MCS) device. The system provides […]
LabCorp Files Patent for Ribosome-Based Microbe Detection Method
According to GlobalData’s company profile on Laboratory Corp of America Holdings, Molecular diagnosis biomarkers was a key innovation area identified from patents. Laboratory Corp of America Holdings‘s grant share as of January 2024 was 44%. Grant share is based on the ratio of number of grants to total number of patents. Microbe detection method using […]
Clearside BioMedical Inc Patent: Method for Retinal Injection
According to GlobalData’s company profile on Clearside BioMedical, Wearable drug delivery devices was a key innovation area identified from patents. Clearside BioMedical‘s grant share as of January 2024 was 34%. Grant share is based on the ratio of number of grants to total number of patents. Method for retinal injection using medical injector Source: United […]
Healthcare inflation forcing US patients to postpone treatments, says study

As the cost-of-living-crisis continues, US patients are worrying about their wallets over their health. Photo: Shutterstock/Gajus. US Healthcare providers (HCPs) fear that inflation is driving patients to postpone or stop treatment, according to a new survey. The GlobalData report, Inflation in Medical Devices, asked healthcare providers across Japan, the US and the five largest Western […]
Spineart completes BAGUERA C IDE trial enrollment in US

In the US, the BAGUERA C prosthesis is under assessments through two separate IDE trials. Credit: Summer Paradive/ shutterstock.com. Spineart has successfully completed patient enrollment in its pivotal US investigational device exemption (IDE) trial for the BAGUERA C cervical disc prosthesis. The BAGUERA C is an investigational device designed to preserve or restore motion and […]
TidalSense launches pilot study for asthma diagnostic device

UK-based respiratory device firm, TidalSense, has announced a new pilot study aimed at evaluating a new device designed to test for respiratory conditions such as asthma in children. Launched in partnership with the University of Nottingham and Nottingham University Hospitals National Health Service (NHS) Trust, the pilot study will examine the use of the N-Tidal […]
