Revvity launches newborn sequencing research workflow

Revvity’s workflow is said to lay the groundwork for future diagnosis of rare diseases in neonates. Credit: Praisaeng/Shutterstock.com. Revvity, a global provider of health science solutions, has unveiled a new ‘sample-to-answer’ workflow designed to accelerate newborn sequencing research. This research-use-only (RUO), flexible end-to-end workflow solution allows users to utilise different reagents, instruments, and databases based […]
DW Healthcare to sell dermoscopy tools manufacturer DermLite

DermLite is engaged in developing advanced dermoscopy equipment, including dermatoscopes and phone adapters. Credit: Yuriy Golub/Shutterstock.com. Private equity company DW Healthcare Partners (DWHP) has announced the sale of DermLite, a developer and manufacturer of dermoscopy tools. DermLite will be sold to EMZ Partners’ portfolio company FotoFinder Systems, a global innovator in skin cancer detection technology. […]
Alithea secures $3.2m to scale RNA sequencing product manufacturing

RNA sequencing can be used to study gene expressions of cells and is used in cancer research, including biomarker discovery, drug resistance, cancer immune microenvironment and immunotherapy. Image Credit: Connect world / Shutterstock. Switzerland-based startup Alithia Genomics has raised SFrF2.8m ($3.2m) in seed funding to scale up the manufacturing capacity of its ribonucleic acid (RNA) […]
Decoding colorectal cancer: prevention in younger populations
In the USA, incident cases among the under 50s have increased by 22% from 2000 to 2013. Credit: Anatomy Image via Shutterstock. March 2024 is Colorectal Cancer (CRC) Awareness Month and it shines a spotlight on a disease that affects millions worldwide. Throughout this dedicated month, individuals, healthcare professionals and organisations join forces to raise […]
Abbott recalls LVAD HeartMate touch communication system
There have been eight injuries and no deaths associated with these disconnections. Credit: ramcreations via Shutterstock. The Class I recall of Abbott’s HeartMate touch communication system involves more than 1,500 devices distributed between 2020 and 2024 and could negatively impact future sales in the left ventricular assist device (LVAD) market. This market was worth $798.6 […]
Scottish university develops diagnostics from chewing gum
Researchers estimate that there are around 16,000 tonnes of plastics produced globally for rapid testing every year to be used in a single-use lateral flow test. Credit: Shutterstock / Ink Drop. Heriot-Watt University has developed a way to turn chewing gum scraped off of the streets of the UK into later flow tests to cut […]
BD to advance bloodstream infections testing in Poland
Sepsis is triggered by BSIs and necessitates swift diagnosis to prevent organ failure, shock, and death. Credit: Becton, Dickinson and Company. Becton, Dickinson and Company (BD) has partnered with Poland’s non-governmental non-profit organisation The Great Orchestra of Christmas Charity (GOCC) to expand testing for bloodstream infections (BSIs) and sepsis management in the country. The collaboration […]
Intuitive’s da Vinci 5 system obtains US FDA 510(k) approval
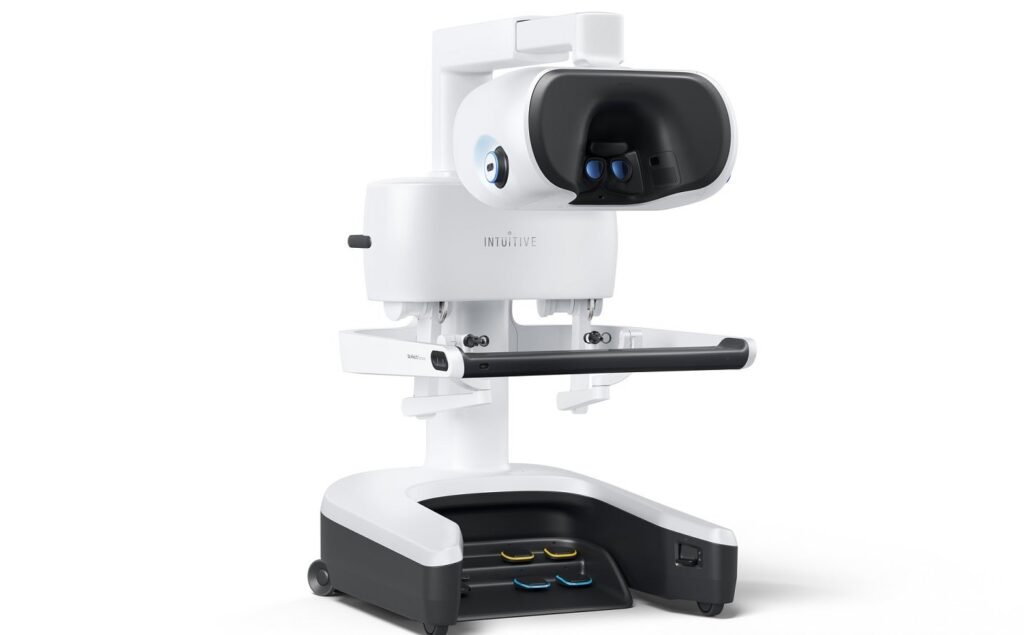
The Intuitive da Vinci 5 surgeon console. Credit: Intuitive Surgical / Globe Newswire. Intuitive has secured 510(k) clearance from the US Food and Drug Administration (FDA) for its fifth-generation multiport robotic system, da Vinci 5. The robotic system builds on the da Vinci Xi, which has been used in more than seven million procedures worldwide. […]
Cosm Medical wins FDA clearance for personalised Gynethotics pessaries

Pelvic organ collapse is very common, with around half of women experiencing it to some degree in their lifetime. Image credit: Shutterstock/Syda Productions. The US Food and Drug Administration (FDA) has granted clearance to Cosm Medical for its Gynethotics Pessaries for pelvic floor disorders. The current gold standard to fit a pessary involves taking manual […]
Bioretec’s spine implant secures FDA breakthrough device status

The Bioretec’s RemeOs Spinal Interbody Cage implant. Credit: Bioretec. The US Food and Drug Administration (FDA) has awarded breakthrough device designation for Bioretec’s RemeOs Spinal Interbody Cage implant, marking a significant advancement in spinal surgery technology. The designation is pivotal for the company’s strategy to introduce the product to the US market. Bioretec’s RemeOs implant […]
