Quibim launches QP-Liver tool for diffuse liver disease diagnosis
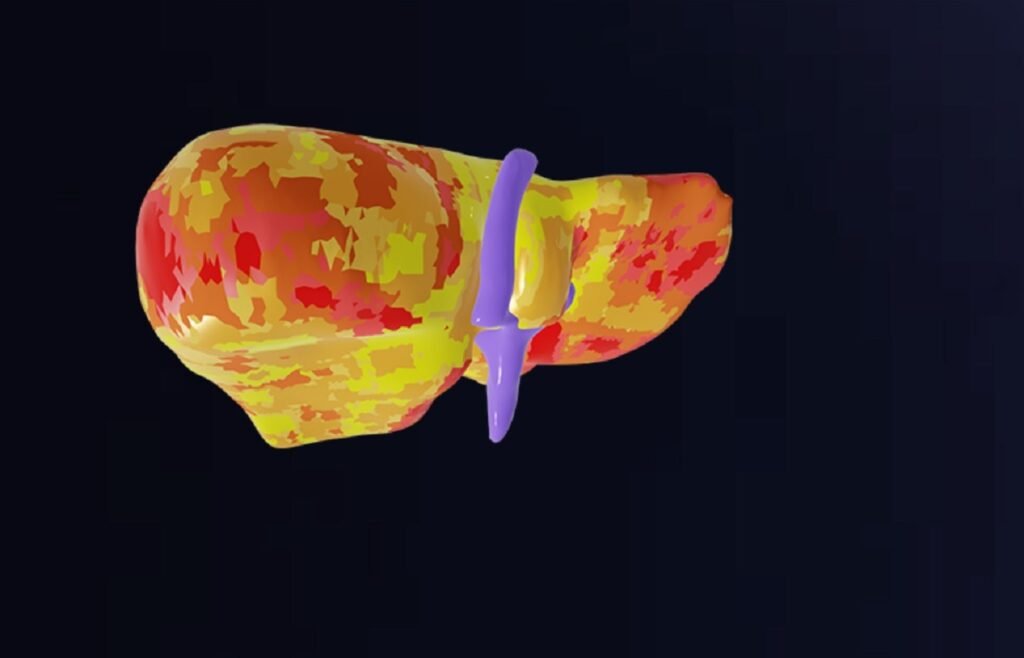
Quilbim’s QP-Liver provides automated liver segmentation and correlating fat and iron quantification. Credit: Quibim. Quibim has announced the launch of QP-Liver, an AI-powered diagnostic tool which is designed to improve the diagnosis of diffuse liver diseases. The tool, which accurately quantifies tissue fat and iron levels from MRI [magnetic resonance imaging] scans, has received clearance […]
Asuragen’s QuantideX diagnostic test secures IVDR certification

The IVDR certification ensures that the test kit meets the stringent requirements set forth for diagnostic tests within the EU. Credit: Motortion Films/Shutterstock.com. Bio-Techne has announced that Asuragen, part of its molecular diagnostics division, has achieved Class C Certification under the European Union In Vitro Diagnostic Regulation (IVDR) for its diagnostic test, QuantideX qPCR BCR-ABL […]
FDA issues 510(k) clearance for RedDrop’s blood collection device

The RedDrop One is a virtually pain-free, low cost, easy-to-use, whole blood and plasma collection and dispensing system. Credit: Innosphere Ventures/ Globe Newswire. The US Food and Drug Administration (FDA) has granted 510(k) clearance to RedDrop Dx’s RedDrop One blood collection device, for prescription use. This approval is a milestone for RedDrop Dx, an Innosphere […]
Precision launches brain–computer interface studies at two sites

The studies plan to enrol up to 15 patients annually at the two sites. Credit: MMD Creative/Shutterstock.com. Precision Neuroscience has expanded clinical research in brain-computer interfaces by launching two new study sites at Mount Sinai Health System and Perelman School of Medicine at the University of Pennsylvania (Penn Medicine) in the US. These studies are […]
FDA tags Vyaire’s recall of over 6.5 million resuscitators as Class I

Vyaire corrected the issue in 2017 but devices distributed before then are still in use today. Image credit: Shutterstock/Tada Images. The US Food and Drug Administration (FDA) has classified the recall of Vyaire’s respiratory support devices as Class I – the most serious type of recall. The US company initiated the recall of its AirLife […]
World Air Quality Report: what are the health impacts and where is it worst?

Dhaka, Bangladesh (pictured) has the second-worst rates of air pollution, and one of the highest concentrations of chronic obstructive pulmonary disease (COPD) in South Asia. Credit: NurPhoto / Getty. Just seven countries had clean air quality in 2023, a new report has found, with analysts pointing to the clear correlation between fine-particle air pollution and […]
UK NICE backs use of GENinCode’s test for ovarian cancer surveillance
The ROCA test will be made available across multiple NHS hospitals across the UK. Image Credit: mi_viri / Shutterstock. The UK National Institute for Health and Care Excellence (Nice) has recommended the use of GENinCode’s Risk of Ovarian Cancer Algorithm (ROCA) test as a preferred test for ovarian cancer surveillance. According to Cancer Research UK, […]
SetPoint joins FDA’s TAP programme with MS device

Multiple sclerosis is caused by the immune system mistakenly attacking the brain and nerves. Credit: Miriam Doerr Martin Frommherz via Shutterstock. After winning breakthrough device designation, SetPoint Medical has been accepted into the US Food and Drug Administration’s (FDA) total product life cycle advisory programme (TAP) for its neuroimmune modulation platform for patients with relapsing-remitting […]
Patent allowed for Bioretec’s RemeOs alloy in US
The RemeOs alloy is said to be a crucial component of Bioretec’s trauma screws. Credit: Roman Zaiets/Shutterstock.com. The US Patent Office has allowed a patent for Bioretec’s RemeOs magnesium alloy composition and will grant and publish it upon company confirmation. The patent encompasses the use of the RemeOs magnesium-calcium-zinc alloy in the manufacturing of orthopaedic […]
Naveris’ NavDx test secures CMS ADLT status
The NavDx blood test is designed for the detection of HPV-driven cancers. Credit: Lightspring/Shutterstock.com. Precision oncology diagnostics company Naveris has received Advanced Diagnostic Laboratory Test (ADLT) status from the Centers for Medicare & Medicaid Services (CMS) for its NavDx test. NavDx is a circulating tumour-tissue–modified HPV (TTMV) DNA blood test developed for the detection of […]
