Vascular Closure Device with Natural & Chemical Agents
According to GlobalData’s company profile on Haemonetics, Glucose monitoring wearbles was a key innovation area identified from patents. Haemonetics‘s grant share as of January 2024 was 53%. Grant share is based on the ratio of number of grants to total number of patents. Vascular closure device using natural and chemical/biological agents Source: United States Patent […]
Glucose Monitoring System with In Vivo and Ex Vivo Components
According to GlobalData’s company profile on DexCom, Medical data analysis was a key innovation area identified from patents. DexCom‘s grant share as of January 2024 was 45%. Grant share is based on the ratio of number of grants to total number of patents. Glucose monitoring system with in vivo and ex vivo portions Source: United […]
Biosense Webster seeks FDA approval for VARIPULSE Platform
The admIRE study’s pilot phase focused on initial device safety and effectiveness. Credit: Peter Porrini/Shutterstock.com. Johnson & Johnson MedTech division Biosense Webster has submitted a Premarket Approval Application (PMA) to the US Food and Drug Administration (FDA) for its VARIPULSE Platform, designed to treat paroxysmal atrial fibrillation. The application is supported by findings from the […]
Metyos raises €2.3m to boost wearable kidney disease device
Metyos has said that its disease management system is able to perform several diagnostic tasks ranging from tracking the impact of diet and medication to spotting warning signs of renal failure. Credit: Shutterstock / Ben Schonewille. French startup Metyos has raised €2.3m ($2.49m) in seed funding for the company’s wearable sensor device, which is designed […]
Abbott’s insertable cardiac monitor receives CE Mark

The device allows patients to continue their daily activities while it monitors heart rhythms. Credit: AnnGaysorn/Shutterstock.com. Abbott has received a CE Mark for its Assert-IQ insertable cardiac monitor (ICM), which is set to provide physicians with a new tool for diagnostic evaluation along with long-term irregular heart rhythm monitoring. The Assert-IQ ICM, part of Abbott’s […]
Novosound’s wearable ultrasound system obtains patent
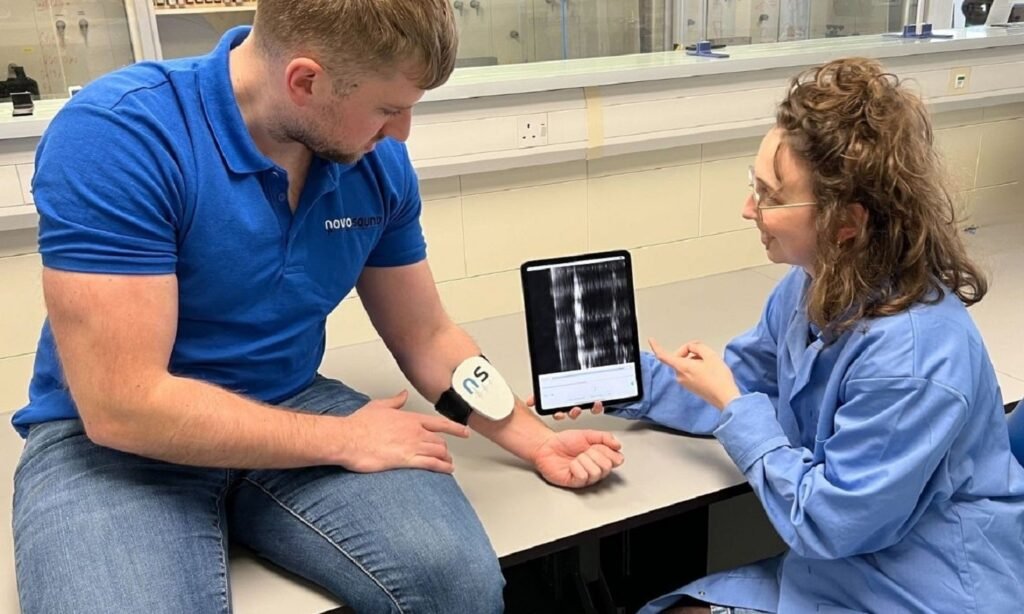
Novosound’s ultrasonic instrumentation system. Credit: Business Wire/Novosound. Scotland-based sensor technology company Novosound has obtained a patent for its wearable ultrasonic instrumentation system. The move comes as the company recently launched its related Slanj platform, which is aimed at the medical and wearable device sectors worldwide. The patent provides Novosound with exclusive rights to market the […]
Neuronetics’s NeuroStar wins FDA clearance for adolescents with depression

The treatment works by stimulating neurons in the brain that regulate mood. Image credit: Shutterstock/dTosh. The US Food and Drug Administration (FDA) has granted clearance to Neuronetics for its NeuroStar device as an add-on therapy to antidepressant therapy in adolescent patients with major depressive disorder (MDD). The transcranial magnetic stimulation (TMS) device is cleared for […]
Castle Biosciences reveals melanoma test study findings at SSO 2024
Melanoma, a type of skin cancer, often presents as a new mole or changes in the appearance of an existing mole. Credit: Africa Studio via Shutterstock. US-based molecular diagnostics company Castle Biosciences has reported positive study data of its gene expression profile (GEP) test, DecisionDx-UM, to predict the risk of sentinel lymph node (SLN) positivity […]
Grindr launches free HIV testing initiative in New Zealand

The announcement comes as part of Grindr’s initiative aimed at expanding LGBT+ access to healthcare services, dubbed Grindr for Equality. Credit: Shutterstock / Vladimka production. Dating app Grindr has partnered with the New Zealand charity the Burnett Foundation Aotearoa to distribute free at-home HIV test kits across the country. The announcement comes as part of […]
Sequana’s DSR therapy shows promise in heart failure patients

The open-label Phase I/II MOJAVE trial (NCT05965934) is designed to evaluate the DRS therapy in patients with congestive heart failure who are resistant to loop diuretic treatment. Image Credit: zentradyi3ell / Shutterstock. Belgium-based Sequana Medical has released data from the Phase I/II MOJAVE study in the US, which is evaluating its direct sodium removal (DRS) […]
