Paragonix commercially launches lung preservation system in US
The BAROguard system received 510(k) clearance from the FDA in August 2023. Credit: mi_viri via Shutterstock. Paragonix Technologies has announced the complete commercial availability of its BAROguard Donor Lung Preservation System in the US. Available to transplant centres and organ procurement organisations across the country, the device is designed to maintain donor lungs in optimal […]
Biolinq raises $58m for glucose microsensor pivotal trial

Biolinq has developed a wearable device that uses an array of electrochemical sensors. Image credit: Business Wire/Biolinq. Biolinq has raised $58m in a financing round to support an ongoing pivotal trial investigating its wearable, non-invasive glucose sensor patch. Biolinq’s CEO Richard Yang told Medical Device Network: “The pivotal study started in March 2024 and we […]
Exact Sciences and Mayo Clinic team up for advanced cancer testing

The data from these tests aims to improve care for individual patients but will also be used to develop advanced AI algorithms, and large language models (LLMs). Credit: Ground Picture via Shutterstock. Exact Sciences and the Mayo Clinic are working together to provide advanced cancer testing to patients, including the OncoExTra and Riskguard tests. This […]
GE HealthCare adds Caption AI to Vscan Air SL
The integration of Caption AI with Vscan Air SL system is in response to challenges posed by cardiovascular disease. Credit: Explode/Shutterstock.com. GE HealthCare has announced the integration of Caption AI technology with Vscan Air SL wireless handheld ultrasound system, aimed at improving cardiac assessments at the point of care. This new feature is set to […]
FDA clears Affinity Biosensors’ LifeScale AST diagnostic platform

LifeScale AST stands out for its rapid results, delivering AST outcomes in under five hours. Credit: Affinity Biosensors. The US Food and Drug Administration (FDA) has granted clearance for Affinity Biosensors’s LifeScale AST system, a diagnostic tool for rapid antibiotic susceptibility testing (AST). This development promises to enhance the management of bacteremia by delivering AST […]
FDA authorises Prenosis’ AI-based tool for sepsis diagnosis
Sepsis is a condition triggered by an overactive response to infection. Credit: Kateryna Kon/Shutterstock.com. The US Food and Drug Administration (FDA) has granted marketing authorisation for Prenosis’ AI tool Sepsis ImmunoScore, to diagnose sepsis. This authorisation, achieved through the De Novo pathway, represents the first-ever FDA approval of an AI/machine learning (ML) software as a […]
France reveals first human brain images from world’s powerful MRI
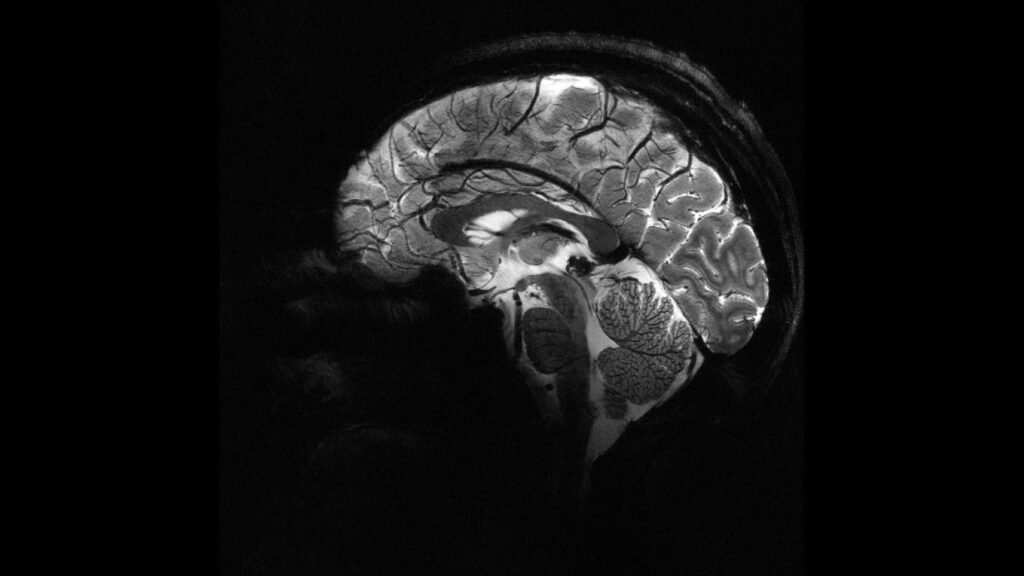
A scan of human brain using the Iseult MRI machine. Credit: © CEA. France’s Atomic Energy Commission (CEA) has revealed the first human brain images captured by the world’s most powerful MRI scanner. The Iseult MRI machine, with a magnetic field strength of 11.7 teslas, represents over two decades of research and development. This milestone […]
Inspira Technologies launches programme examining artificial womb

Last month, Inspira had applied for approval for its INSPIRA ART100 device from the Israeli Government’s Ministry of Health medical device regulation unit. Credit: Shutterstock / Iryna Inshyna. Israeli respiratory medical device company Inspira Technologies has announced the launch of a programme designed to explore the use of its INSPIRA ART100 device, an artificial womb […]
Kardium’s pulsed field ablation clinical trial enters final stages

Kardium’s device is called the Globe System – a 122 gold electrode catheter that delivers pulsed field energy to the heart. Image credit: Business Wire/Kardium. Kardium has completed enrolment in a study evaluating its pulsed-field ablation (PFA) device for the treatment of atrial fibrillation. According to a ClinicalTrials.gov entry, the PULSAR trial (NCT05462145) is estimated […]
Kelyniam, Finceramica announce FDA clearance for NEOS system
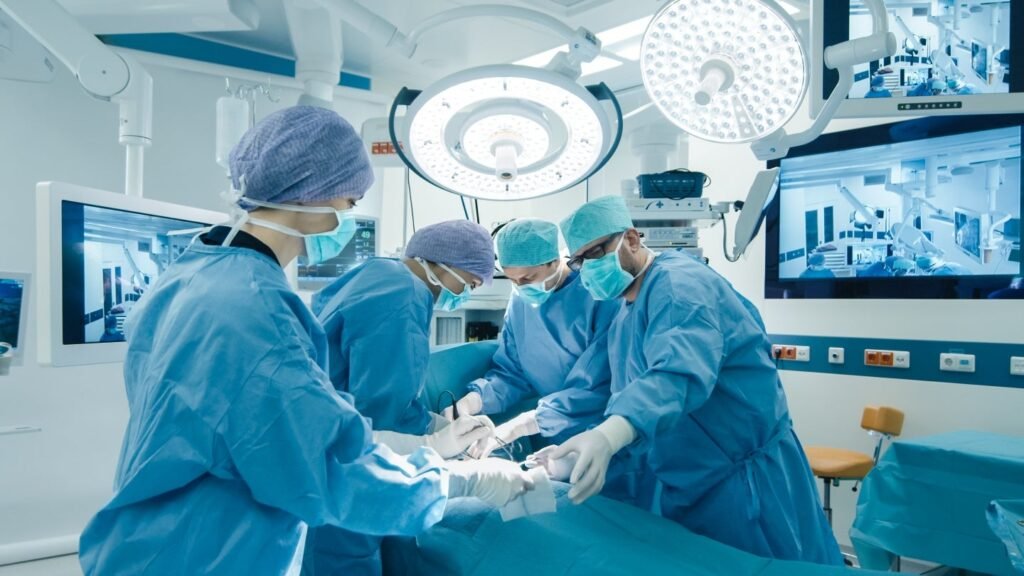
More than 9,000 CustomizedBone implants have been integrated into patients. Credit: Gorodenkoff/Shutterstock.com. Kelyniam Global and Finceramica Faenza have announced the Food and Drug Administration (FDA) 510(k) clearance for the NEOS Surgery Cranial LOOP fixation system to be used with the latter’s CustomizedBone hydroxyapatite cranial implant. The Cranial LOOP system, crafted from PEEK-OPTIMA, a biocompatible polymer, […]
