Biotechs must prioritise ethics to deter GenAI “bad actors”

At the ongoing London Biotechnology Show, Thomas Balkizas, Microsoft’s senior director of life sciences, highlighted the importance of maintaining ethical practices in AI development. Credit: Bloomberg via Getty Images. AI developers in the biotechnology field need to watch out for potential “bad actors”, who could use the technology for malicious purposes, says Thomas Balkizas, the […]
Hope for Parkinson’s patients: The brain implants changing lives

Technology is making dramatic improvements to patients living with Parkinson’s disease. Credit: Kohji Asakawa / DeltaWorks via Pixabay. Advanced devices are now giving hope to greater numbers of patients living with Parkinson’s disease. Millions of people worldwide are affected by the chronic and progressive neurological disorder. The disease predominantly affects the motor system, leading to […]
Leica Microsystems adds Viventis Microscopy to portfolio

Leica Microsystems will provide worldwide support and service for all Viventis Microscopy products from now on. Credit: Shutterstock/Aris-Tect Group. Leica Microsystems, a subsidiary of medical device conglomerate Danaher, has added Viventis Microscopy to its portfolio. The company’s Viventis LS2 Live Dual View Light Sheet Fluorescence Microscope uses a patented combination of dual illumination, dual view […]
Qiagen and FBI partner on DNA quant digital PCR assays
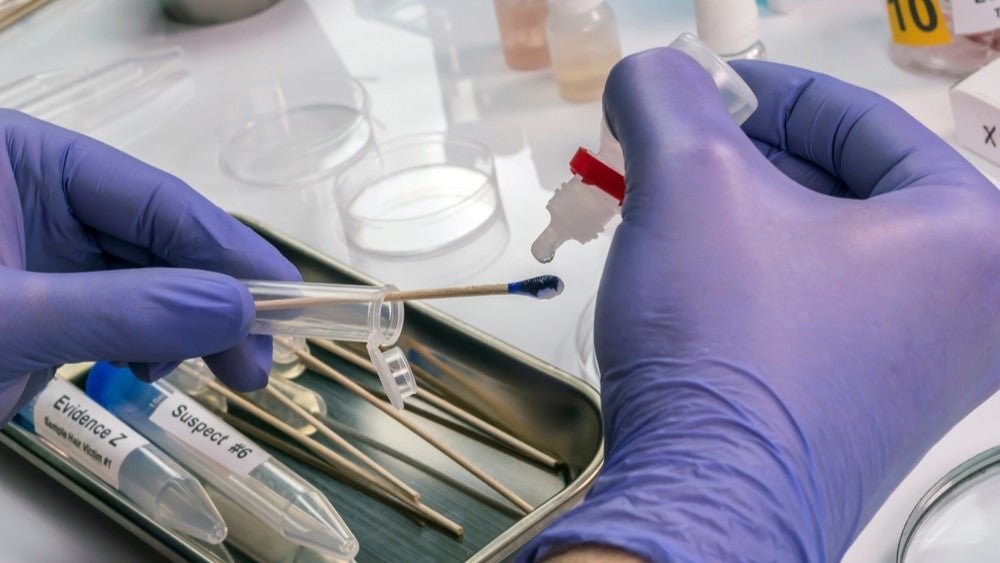
The new digital PCR assay developed as part of the Qiagen and FBI collaboration will be run on the former’s QIAcuity digital PCR devices. Image Credit: felipe caparros / Shutterstock. The US Federal Bureau of Investigation (FBI) is partnering with Netherland-based Qiagen to develop a novel assay capable of detecting and quantifying minimal amounts of […]
Averto Medical secures $30.5m to advance ColoSeal system
The system could potentially benefit over 100,000 patients each year. Credit: MAD.vertise via Shutterstock. Averto Medical has closed an oversubscribed $30.5m Series A financing round, which will help advance its ColoSeal medical device that avoids the need for ostomy. The funding round was led by Cormorant Asset Management and supported by Venrock Healthcare Capital Partners, […]
MHRA report finds wellbeing apps welcomed but users wary of AI chatbots

The MHRA say that the key findings of its report include the development of a conceptual framework for categorising DMHTs as well as considerations for regulation and evaluation. Credit: Shutterstock / Rawpixel qThe UK’s Medicines and Healthcare products Regulatory Agency (MHRA) has published initial findings from a three-year report intended to set the groundwork for […]
FDA approves CereVasc’s eShunt system study for hydrocephalus
The eShunt System provides a percutaneous transvenous-transdural access to the central nervous system. Credit: Thomas Design/Shutterstock.com. The US Food and Drug Administration (FDA) has granted investigational device exemption (IDE) approval for CereVasc to initiate the STRIDE pivotal study of its eShunt system for treating normal pressure hydrocephalus (NPH). This study will compare the eShunt system’s […]
Psilocybin effective in treating depression

Psilocybin found in mushrooms (often known as ‘magic’ mushrooms) has shown potential in treating depression. Credit: YARphotographer / Shutterstock A new study published in the British Medical Journal found potential in the use of psilocybin to treat depression, with the greatest improvements seen in older patients, those with secondary depression and patients who had previously […]
Samsung agrees to acquire foetal ultrasound AI company Sonio
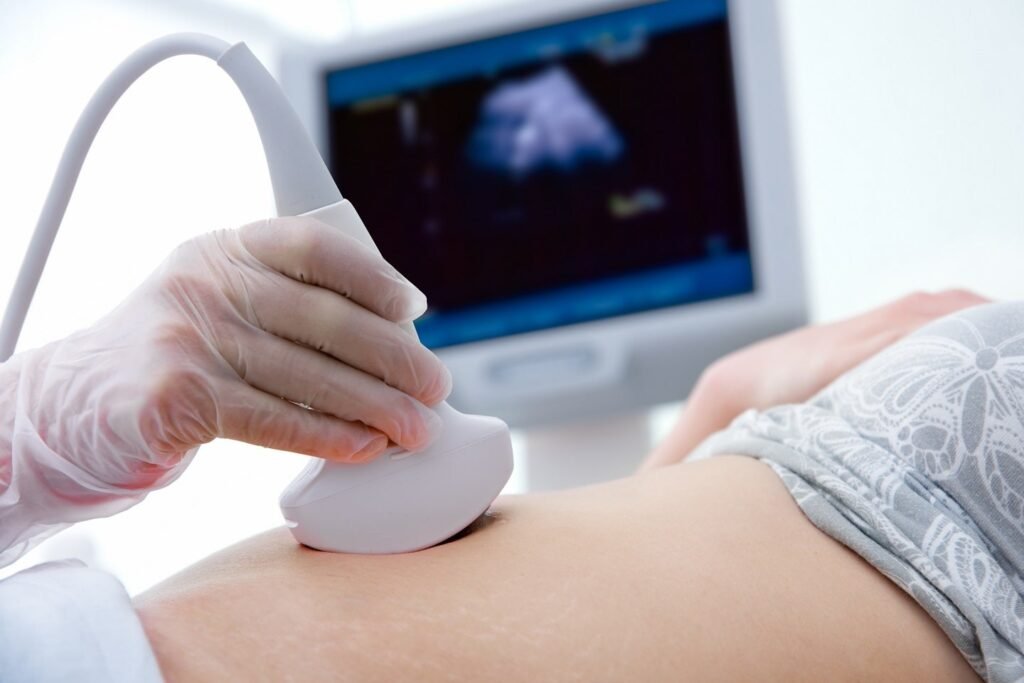
Samsung Medison intends to advance AI in ultrasound technology with the acquisition of Sonio. Credit: Alexander Raths/Shutterstock.com. Samsung Medison, an affiliate of Samsung Electronics and a global medical equipment company, has entered an agreement for the acquisition of France-based foetal ultrasound AI software company Sonio. The deal is valued at about $92.4m, reported Reuters. The […]
FDA issues 510k clearance for US Medical’s ablation system
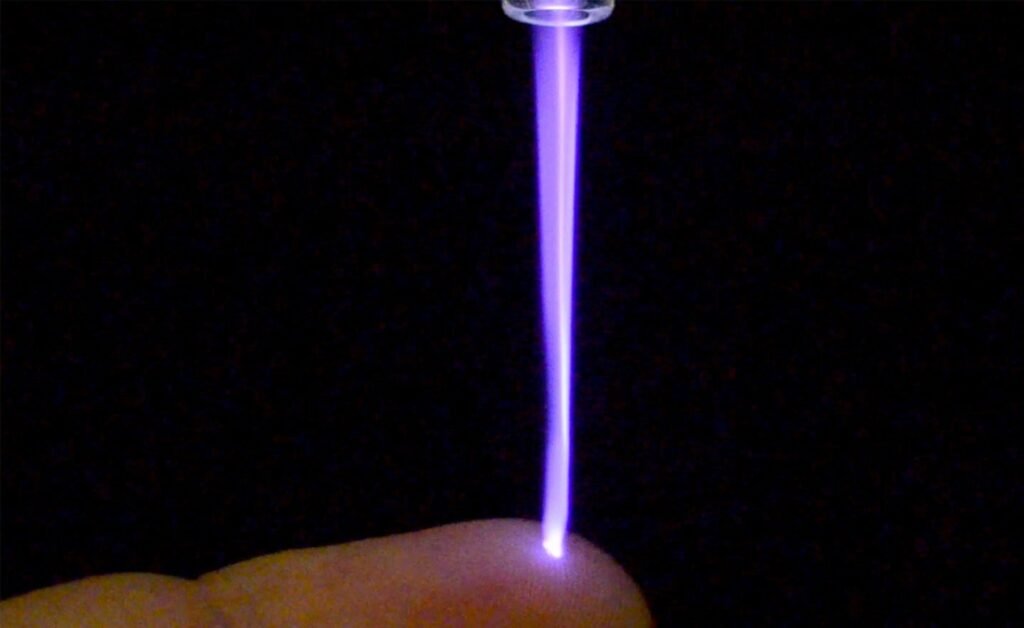
US Medical Innovations’s Canady Helios Cold Plasma System. Credit: Business Wire/ US Medical Innovations, LLC (USMI). The US Food and Drug Administration (FDA) has issued 510k clearance for US Medical Innovations’s Canady Helios Cold Plasma (CHCP) Ablation System, a new technology designed for the ablation of soft tissue at the time of surgery. The system […]
