Rapid Medical receives approval for revascularisation device
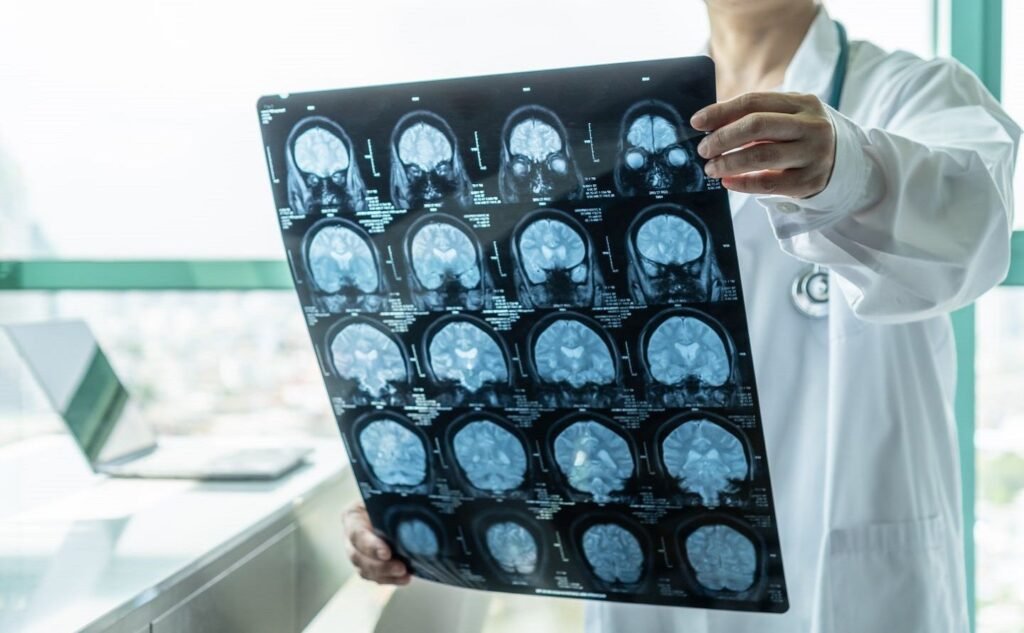
The device helps remove blood clots from the brain to advance ischemic stroke treatment. Credit: Chinnapong / Shutterstock.com. Rapid Medical has secured approval from China’s National Medical Product Administration (NMPA) for its TIGERTRIEVER™ revascularisation device. Claimed to be the world’s only adjustable thrombectomy device, TIGERTRIEVER provides patient-specific solutions to remove blood clots from the brain […]
Cardinal Health introduces new medical device for newborn infants

The device helps newborns and infants born prematurely to develop their non-nutritive sucking skills. Credit: tomkawila / Shutterstock.com. Medical and laboratory products provider Cardinal Health has introduced its next-generation medical device, NTrainer System 2.0, for premature and newborn infants. Designed for improving neonatal feeding, the NTrainer System 2.0 is claimed to be the only US […]
FDA approves AION’s iTempShield device and software system

The device can be used in remote patient monitoring environments, hospitals. Credit: Shyntartanya / Shutterstock.com. The US Food and Drug Administration (FDA) has granted 510 (k) clearance for AION Biosystems’ iTempShield device and software system. The quarter-sized, skin-wearable device iTempShield leverages cloud-based software and proprietary algorithms and is intended for adults, and children aged five […]
Urotronic reveals follow-up data for its Optilume BPH catheter system
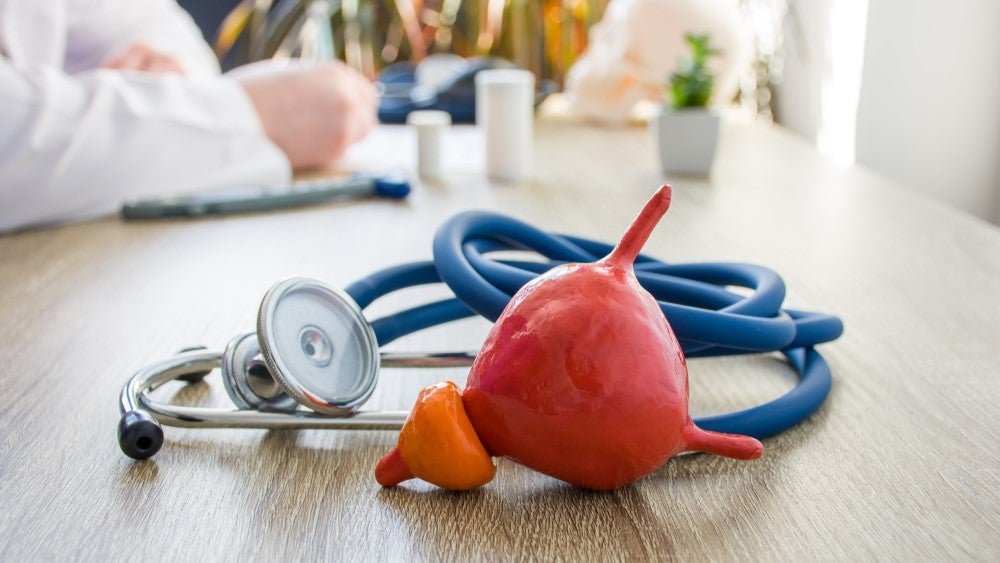
The randomised clinical trial (NCT04131907) evaluated the efficacy and safety of the drug-device system over 12 months in 148 patients with LUTS secondary to BPH. Image Credit: Shidlovski / Shutterstock. One-year follow-up data from a clinical trial evaluating Urotronic’s Optilume BPH Catheter System has been published in the peer-reviewed American Urological Association journal, Journal of […]
Epineuron enrols first patient in nerve regeneration study
In the trial, the FDA breakthrough designated device will deliver electrical stimulation as an hour-long, single dose near the site of nerve injury. Image credit: Shutterstock/Giovanni Cancemi. Epineuron has enrolled the first patient in a trial investigating its PeriPulse device for the treatment of acute peripheral nerve injury. The Ontario, Canada-based company plans to enrol […]
FDA clears Daye’s gamma-irradiated Nude tampon

Daye’s founder and CEO Valentina Milanova said the FDA greenlight “marks a pivotal moment in our mission to enhance the health and well-being of women and assigned female at birth individuals.” Image credit: Daye. UK gynaecological startup Daye will be able to reach women’s and girls’ health across the pond, after the US Food and […]
FDA identifies Hamilton Medical ventilator recall as Class I

Four models of Hamilton Medicals ventilators have been recalled after reports of software issues causing the devices to switch to Ambient State Mode. Credit: HVEPhoto/Shutterstock Ventilator manufacturer Hamilton Medical has launched a voluntary recall of four of its ventilators following reports of software troubles that cause the device to stop working without warning. The products in […]
ANVISA grants marketing approval to Zylox-Tonbridge’s catheter
The catheter’s tapered tip and short shoulder enables easier access through complex lesions. Credit: Dmitry Kalinovsky/Shutterstock.com. Brazilian Health Regulatory Agency ANVISA has granted marketing approval to Zylox-Tonbridge Medical Technology’s ZENFlow Tiger LD PTA Dilatation Catheter (ZENFlow Tiger). With this approval, the company intends to launch the product in the South American market. ZENFlow Tiger features […]
Medical Device Network Excellence Awards 2023: Cydar Medical

Cydar Medical is a Cambridge, UK, based health-technology company whose integrated software solution enables hospitals to access the full potential of AI based clinical applications and data insights. Founded in 2012, the Company is on a mission to advance the quality of care to patients globally. It is dedicated to partnering with healthcare organizations to […]
Quest Diagnostics’ AAV test receives breakthrough designation

Quest will also provide Sarepta with clinical lab testing. Credit: Rapeepat Pornsipak / Shutterstock.com. Quest Diagnostics has secured breakthrough device designation for its adeno-associated virus (AAV) test, AAVrh74 ELISA assay, from the US Food and Drug Administration (FDA). The test is an enzyme-linked immunosorbent in vitro diagnostic assay intended for the semi-quantitative detection of antibodies […]
