Vibrant Wellness introduces new Toxin Genetics Test
The new test helps assess specific genetic biomarkers linked to the body’s metabolic detoxification process. Credit: Tatiana from Pixabay. Vibrant Wellness has expanded its functional lab test portfolio with the launch of the new Toxin Genetics Test for personalised detox protocols. The new test has been developed for evaluating specific genetic biomarkers linked to the […]
Biomerics completes Brooklyn Park facility expansion in US

Biomerics’s facility expansion includes 20,000ft² of space for micro metals laser processing. Credit: Biomerics. Medical device contract manufacturer Biomerics has completed a 100,000ft² expansion of its operations in Brooklyn Park, Minnesota, US. With the expansion, the facility now has a total area of 170,000ft². This investment enables the consolidation of multiple facilities and expands the […]
How pharmaceutical manufacturers can guarantee a seamless eIFU implementation

Getting an eIFU platform online always comes with its fair share of challenges – but working with the right partner can make it a breeze. Implementing an eIFU-platform is a significant undertaking. But the potential benefits more than making up for this; IFUs can be made available on websites as stipulated in Medical Device Regulations […]
Shedding light on Brain Cancer Awareness Day and Brain Tumor Awareness Week in Canada
Brain tumours pose a significant health risk as they can put pressure on areas of the brain that are used for thought, emotion, and movement. Credit: Dmytro Zinkevych via Shutterstock. Brain Cancer Awareness Day, which occurs in Canada on 24 October, and Brain Tumor Awareness Week, which occurs globally during 28 October – 4 November, […]
Medtronic’s Simplera continuous glucose monitor receives CE mark
The CGM device market is booming and GlobalData forecasts that CGMs will generate $8.4bn in sales by 2030. Image Credit: Andrey_Popov / Shutterstock. Medtronic has received a CE mark for its continuous glucose monitoring (CGM) device, Simplera, allowing the device to be marketed in the European Union. The company will begin a phased launch of […]
Realeve to provide neuromodulation system for post-stroke study
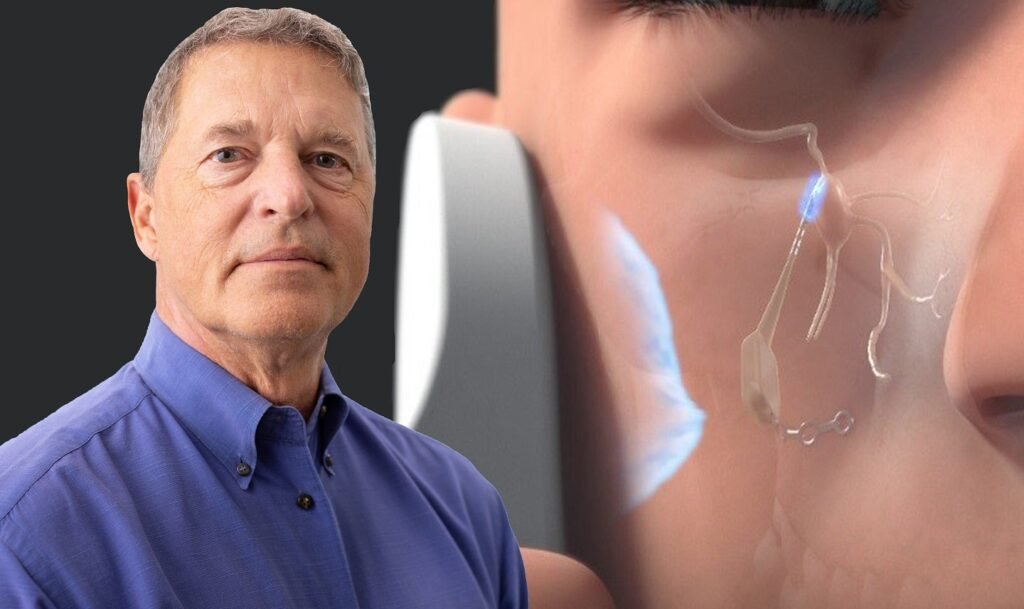
Realeve founder Peter Bonutti has introduced the breakthrough micro implant for the treatment of stroke, Alzheimer’s and other brain diseases. Credit: Realeve/Business Wire. Realeve has secured a grant to provide its Pulsante micro-neuromodulation system for a study aimed at advancing post-stroke recovery. The study will be conducted at the Cleveland Clinic. It will use the […]
Signal: CMR Surgical Raises $165m to continue development of surgical robot

CMR Medical has now surpassed $1bn in capital funding. Photo: T. Schneider / Shutterstock.com. Cambridge, UK-based CMR Surgical has raised $165m in funding to continue the development of its Versius surgical robot. This latest investment brings their total raised to over $1bn, cementing the coveted unicorn status the company acquired in 2019 through a £195m […]
Tyber Medical receives approval for K-Wires and Steinmann Pins
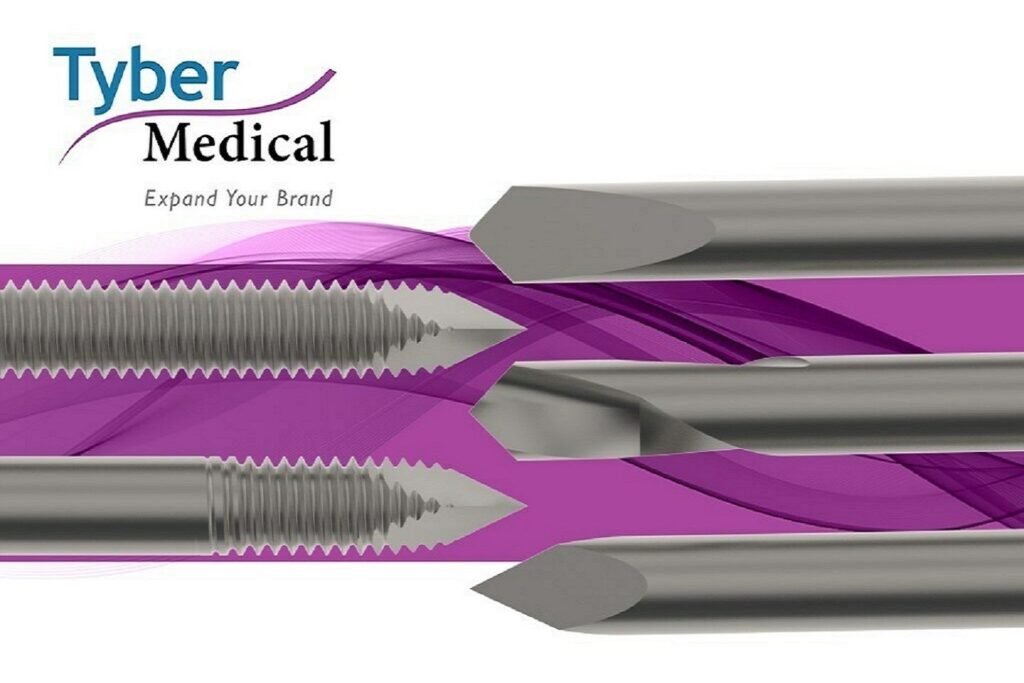
The cleared K-Wires and Steinmann Pins are available in both sterile and non-sterile options and come in multiple configurations. Credit: Tyber Medical LLC/ PRNewswire. Tyber Medical has secured approval from the US Food and Drug Administration (FDA) and European Union Medical Device Regulation (EU MDR) for its implantable K-Wires and Steinmann Pins. Both the wires […]
Exactech’s Activit-E polyethylene receives US FDA clearance

Exactech has received FDA clearance for Activit-E polyethylene for the Vantage total ankle system. Credit: Exactech / Business Wire. Exactech has received 510(k) clearance from the US Food and Drug Administration (FDA) for its Activit-E polyethylene for the VantageTotal Ankle System. Last month, the company also obtained clearance from the FDA for Activit-E for the […]
Riverside University Health System to deploy FloPatch technology
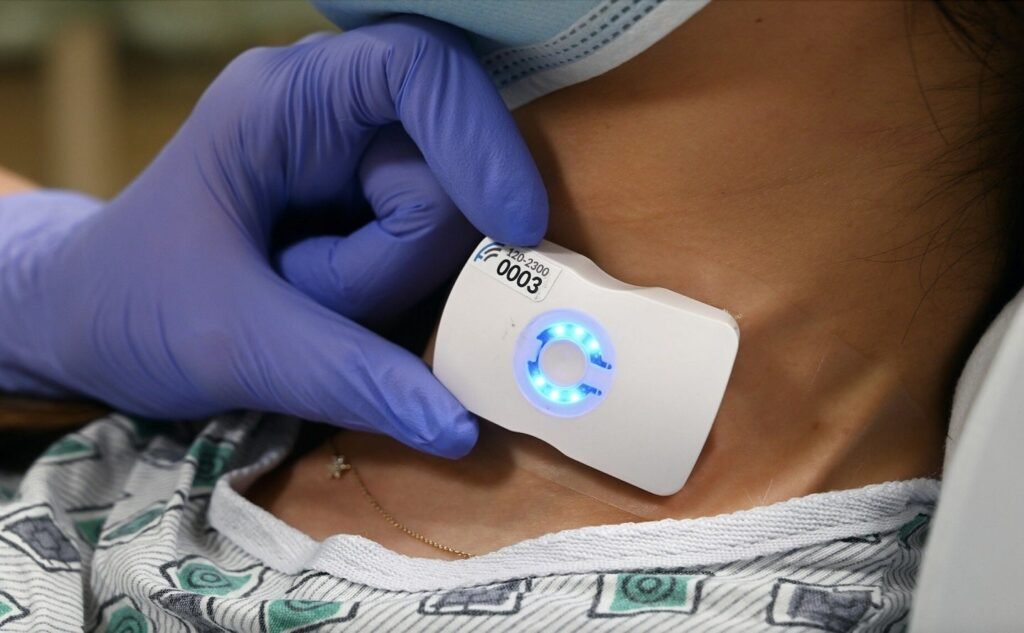
FloPatch will help precisely evaluate a patient and determine the most suitable therapeutic intervention. Credit: Flosonics Medical / Business Wire. Riverside University Health System has announced the deployment of Flosonics Medical’s FloPatch medical technology for fluid management in sepsis. Approved by the US Food and Drug Administration (FDA), the wireless, wearable tool enables real-time precision […]
