UK launches national full-body scan platform to accelerate drug discovery
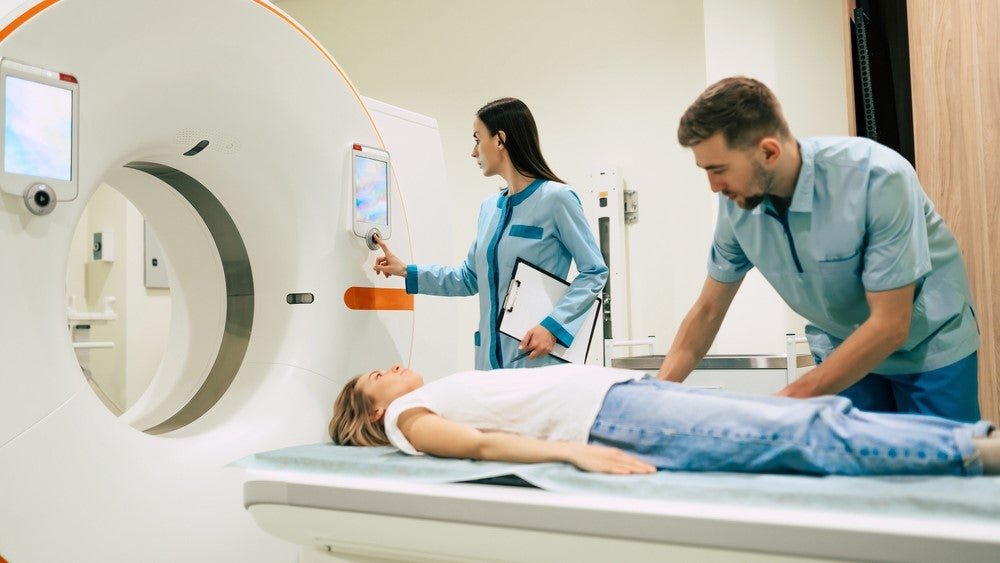
The project received £32m ($38.8m) in funding from the UK government through the UK Research and Innovation (UKRI) Infrastructure Fund. Image Credit: My Ocean Production / Shutterstock. The UK national scanning programme, National PET Imaging Platform (NPIP), has been launched to conduct total-body positron emission tomography (PET) imaging to accelerate drug discovery. The NPIP was […]
Sanguina receives US FDA clearance for haemoglobin test kit
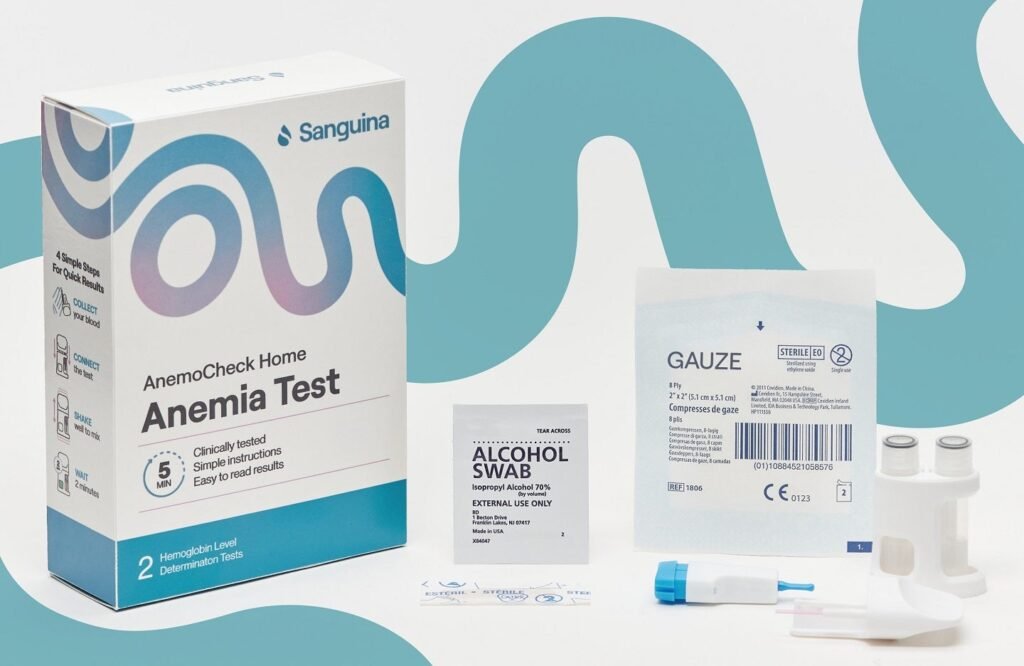
AnemoCheck Home is the only FDA-cleared home anaemia test with instant results. Credit: Sanguina/Business Wire. Sanguina has received clearance from the US Food and Drug Administration (FDA) for its haemoglobin test kit, AnemoCheck Home. Said to be the only FDA-approved home haemoglobin test kit in the US, the test enables individuals with anaemia due to […]
Aventa Genomics introduces NGS test for solid tumours

Aventa FusionPlus test helps identify gene fusions, translocations and rearrangements across 361 genes from FFPE tumour tissue. Credit: Arek Socha from Pixabay. Aventa Genomics has launched its next-generation sequencing (NGS) test, Aventa FusionPlus, to identify gene fusions and translocations, along with rearrangements across 361 genes from formalin-fixed, paraffin-embedded solid tumour tissue. The test incorporates 3D […]
MD&M 2023: Discover medtech innovations at Advanced Manufacturing Minneapolis

Advanced Manufacturing Minneapolis is the Midwest’s largest annual end-to-end design and manufacturing event, combining five connected expos in one epic event: MD&M, Automation Technology Expo (ATX), Design & Manufacturing (D&M), MinnPack and Plastec. Minnesota is a renowned healthcare hub, home to Mayo Clinic, the top-ranked hospital in the US, as well as the largest private […]
FDA launches neurological devices in TAP pilot

The FDA are advancing the pilot.. Credit: grandbrothers via www.shutterstock.com The US Food and Drug Administration (FDA) is expanding the Total Product Life Cycle Advisory Programme (TAP) to neurological and physical medicine devices, to accelerate device development in neurological and physical medicine technologies. The aim of the programme is to supply US patients with high-quality […]
Amber Implants commences first-in-human trial for vertebral fracture device
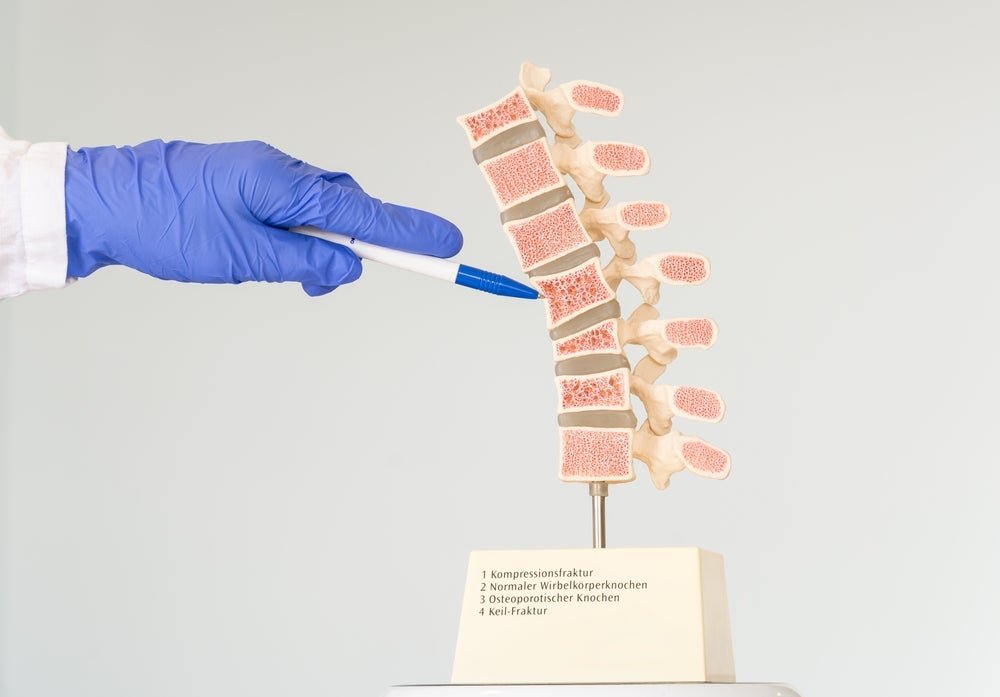
The Hague, Netherlands-based medtech company has designed its system for both single and multi-level posterior fixation. Image credit: Shutterstock/Lilia Solonari. Amber Implants has kickstarted a clinical trial investigating the safety and effectiveness of its implant designed to treat vertebral compression fractures (VCF). The first-in-human study will test the company’s VCFix spinal system implant and is […]
Palantir set to win UK National Health Service overhaul contract

Palantir are mostly known for their work in the US defense sector. Photo: Fabrice Coffrini/AFP via Getty Images. Palantir, Peter Thiel’s data analytics and defence company, are likely to be announced in the near future as the winners of a £480m ($582m) contract to overhaul the National Health Service (NHS) of the UK. Bloomberg reported […]
Microsure raises $40m for microsurgical robot
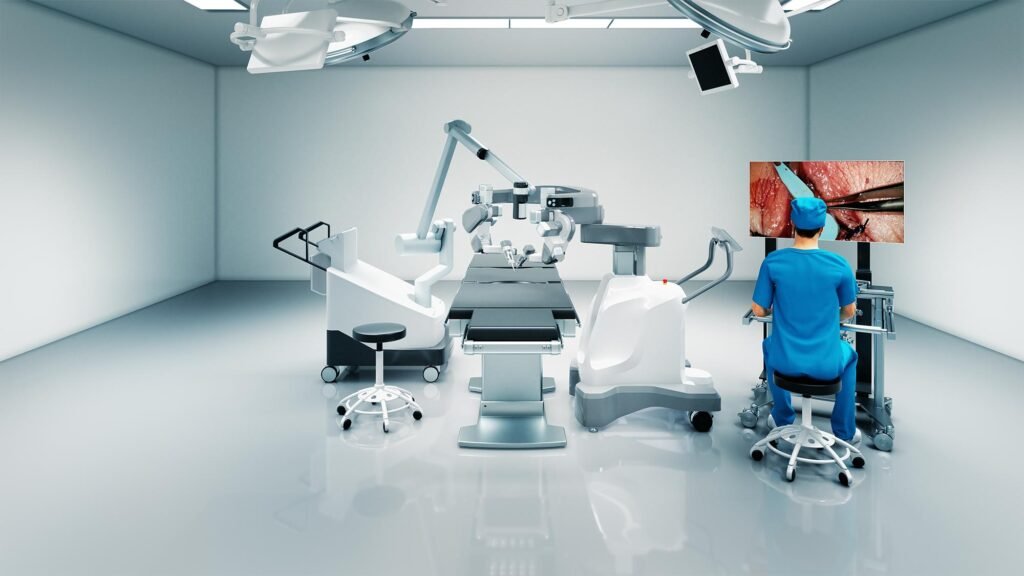
A rendering of the MUSA-3 robot with surgeon console. Image credit: Microsure. Microsure has closed a Series B2 investment round worth $40m (€38m). The company stated it will use the funding to advance its microsurgical robot MUSA-3 through clinical studies and regulatory clearings. The European Innovation Council Fund (EICF), kineo, and invest-NL join existing shareholders […]
Akura Medical raises funds for mechanical thrombectomy platform
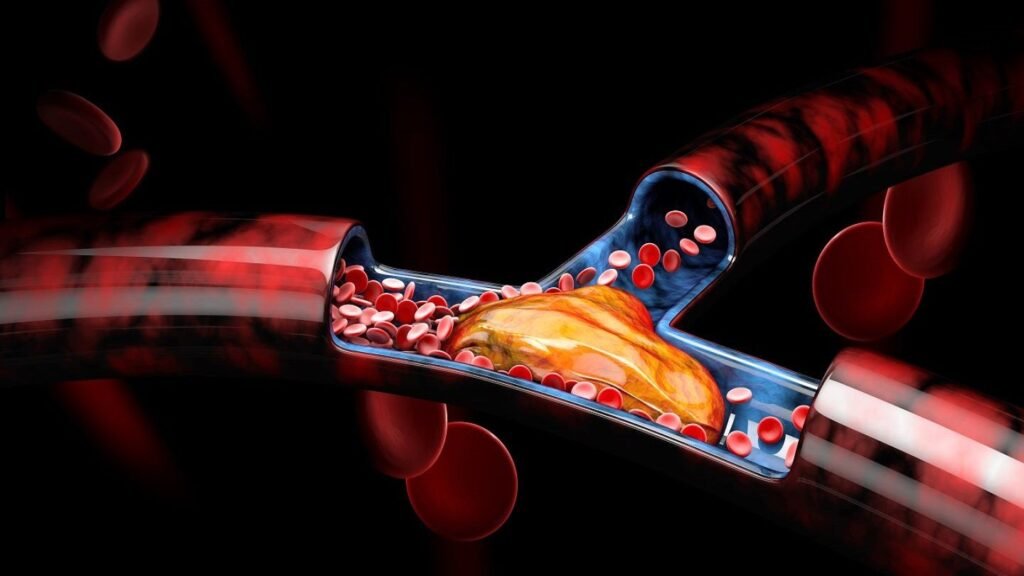
The Akura platform is designed to help remove soft and hard clots. Credit: Victor Josan/Shutterstock.com. Akura Medical, a Shifamed portfolio company, has closed its Series B financing round, through which it raised $35m in funding. The funds will be used to apply for US Food and Drug Administration (FDA) 510(k) approval for its mechanical thrombectomy […]
ProciseDx obtains FDA de novo clearance for Humira TDM tests

A ProciseDx analyser and assay. Credit: ProciseDx Inc/PRNewswire. ProciseDx has obtained de novo clearance from the US Food and Drug Administration (FDA) for its therapeutic drug monitoring (TDM) tests for drugs adalimumab (Humira and biosimilar Amgevita) and infliximab (Remicade and biosimilars Inflectra and Renflexis). Procise ADL and Procise IFX assays are intended for quantifying adalimumab (ADL) and infliximab (IFX) in patients suffering with inflammatory bowel diseases (IBD). Designed to be performed using serum, […]
