Medical Device Network Excellence Awards 2023: Nuova SB System

Based in Milan, Nuova SB System is a provider of Endoscope Reprocessing and Filtration Solutions including Treatment Systems, Disinfection, Purification and Sterilization. The company is a Category Award Winner for Innovation and Product launches in the 2023 Medical Device Network Excellence Awards for the Launch of new products incorporating monitoring technology in the form of […]
DELFI Diagnostics introduces FirstLook Lung blood test
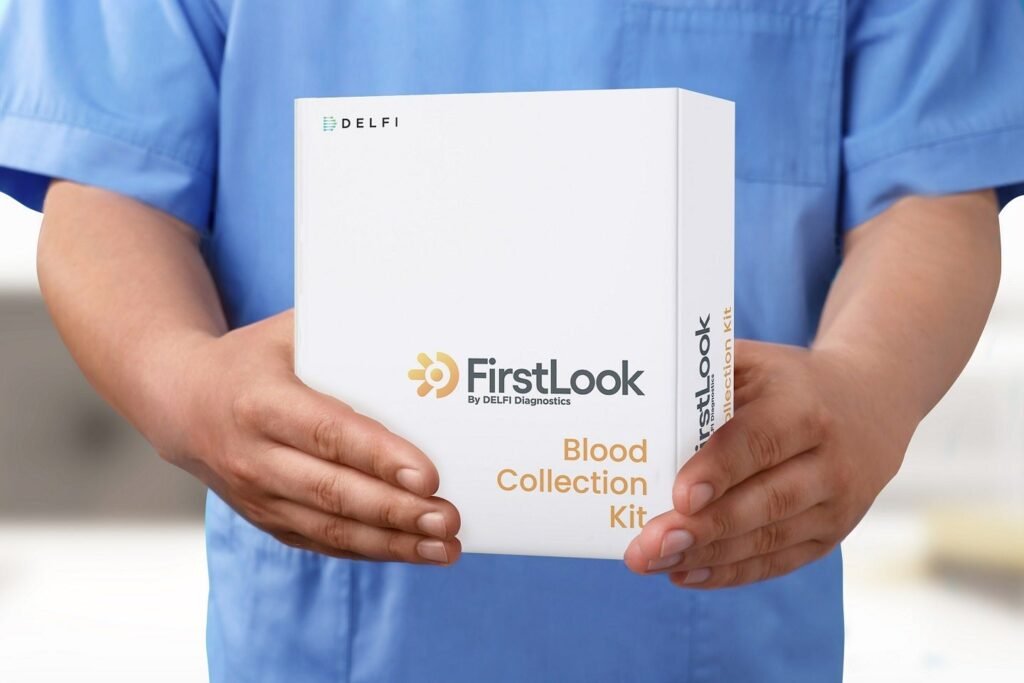
FirstLook Lung is a blood test that helps determine the likelihood of detecting lung cancer through a low-dose CT scan. Credit: DELFI Diagnostics/ PRNewswire. DELFI Diagnostics has announced the commercial availability of its new blood test, FirstLook Lung. The blood test has been designed to determine the likelihood of identifying lung cancer through low-dose CT […]
Tento+ debuts a generative AI tool to support medical device compliance

Tento+ also provides design requirements and test plan suggestions, along with tailored insights into these fields. Image Credit: NicoElNino/Shutterstock. AND Technology Research has launched a generative artificial intelligence (AI) dashboard update to its medical compliance tool, Tento+. The new update is designed to streamline the medical compliance process. As per AND Technology, the Tento+ AI […]
HLTH 2023: Google Cloud taps into GenAI health market with Vertex AI

2023 has been dubbed as generative AI’s “breakout” year and tech giant Google is flexing its muscles once again in the healthcare space. At the HLTH 2023 conference this week, Google Cloud announced updates to its Vertex AI Search engine for healthcare and life sciences organisations. The new feature enables medically tuned generative AI powered […]
HLTH 2023: Urgency needed to address neurodegenerative disease space

The annual HLTH conference convenes this week in Las Vegas from Oct 8-11. Image credit: Catherine Longworth As the prevalence of neurodegenerative diseases rapidly rises, researchers at the ongoing HLTH 2023 conference highlighted how biomarker development is urgently needed for treatment breakthroughs to occur. In the United States, as many as 6.2 million people may […]
enVVeno Medical unveils plans to expedite enVVe development
The enVVe valve is designed to accommodate the natural dilation and contraction of the vein. Credit: Victor Josan/Shutterstock.com. enVVeno Medical has unveiled a new expedited development plan for its transcatheter-based replacement venous valve, enVVe. The new plan is anticipated to help lower the development cycle of the valve by roughly six months. Expected to start […]
Study calls for smartphone treatment for pregnant diabetes patients

Researchers for the study trialed Hybrid Closed-Loop or Artificial Pancreas technology as part of the study carried out at University of East Anglia. Credit: Shutterstock / Alina Troeva The UK’s National Institute for Health and Care Research (NIHR) is calling for smartphone-delivered diabetes treatments to be offered to pregnant women suffering from diabetes following a […]
Hemodynamic monitoring module secures EU MDR CE Mark
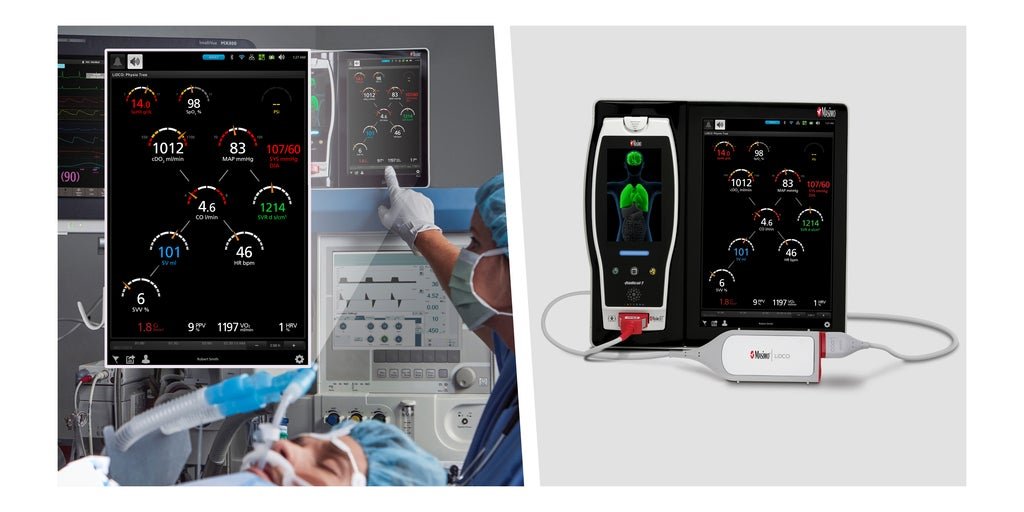
The LiDCO BIC module is designed to connect to multi-patient monitoring platforms, including Masimo Root Patient Monitoring and Connectivity Hub, to provide advanced hemodynamic monitoring. Credit: Business Wire Medical technologies company Masimo has just received the EU Medical Device Regulation (MDR) CE mark for its LiDCO board-in-cable (BIC) module designed to connect to multi-patient monitoring […]
Signal: LimmaTech raises $37m for antimicrobial resistance research

Franz-Werner Haas, LimmaTech CEO. Photo: Thomas Kienzle/AFP via Getty Images. Limmatech, the GSK spinout focusing on antimicrobial resistance, announced on Monday (9 October) that it has raised $37m in a series A funding round. The round was co-led by Adjuvant Capital, AXA IM Alts and Novo Holdings, the parent company of obesity drugs giant Novo […]
Sigrid starts clinical trial for diabetes and weight control device

The SiPore21 device, if cleared, will be registered as a Class IIb medical device. Image Credit: Mikhailava Alesia / Shutterstock. Sigrid Therapeutics has started the Shine clinical trial for SiPore21, the device designed to control diabetes and obesity. SiPore technology is based on mesoporous silica particles (MSPs), a colloidal amorphous silicon dioxide matrix. It acts […]
