price change in recent years
The selling price or list price of a medical device is driven by numerous factors, such as therapy area and complexity of procedure, level of reimbursement, development costs, the number of competing manufacturers, product life cycle, and whether there have been any recent new technology or product launches. In addition, the final selling pricing of […]
Masimo gets de novo approval from FDA for ORi
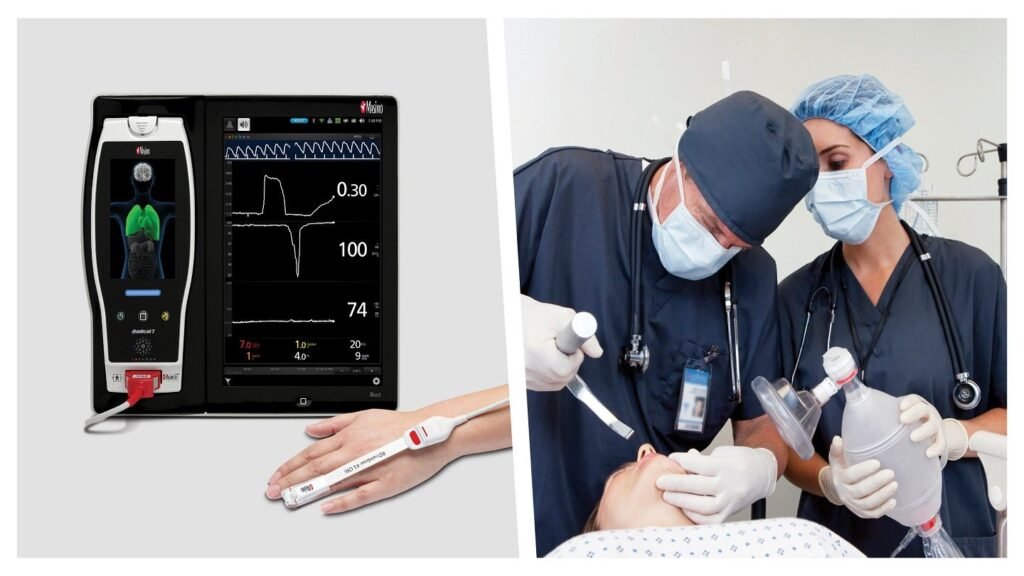
Masimo’s Ori helps deliver additional insight into a patient’s oxygen status in the moderate hyperoxic range under supplemental oxygen. Credit: Masimo / Business Wire. Masimo has received de novo approval from the US Food and Drug Administration (FDA) for ORi, its noninvasive, continuous parameter that delivers additional insight into a patient’s oxygen status in the […]
SOTIO sign ADC platform licencing agreement with Synaffix
The licensing agreement between SOTIO and Synaffix will see it leverage ADC technology platform combined with proprietary antibodies to target solid tumours. Credit Jose Luis Calvo/Shutterstock Biotechnology company, Synaffix which was acquired in June 2023 by Lonza, will collaborate with clinical-stage immuno-oncology company SOTIO in a new licensing agreement to leverage antibody-drug conjugates (ADCs) technology […]
Illumina vows to challenge European Commission order to give up GRAIL

Illumina has been ordered to divest itself of GRAIL after the European Commission feared it would limit customer choice. Credit: Shutterstock / Vitalii Vodolazskyi Illumina has said that it intends to challenge the European Commission’s (EC) ruling calling for the company to divest itself of its newly acquired cancer detection firm, Grail. The San Diego-based […]
NMPA approves Geneseeq’s NSCLC TMB Kit
The kit qualitatively detects TMB in FFPE tissue samples of patients with EGFR/ALK-negative non-squamous NSCLC. Credit: 15Studio/Shutterstock.com. The Chinese National Medical Products Administration (NMPA) has granted approval as a breakthrough medical device for Geneseeq Technology’s Non-Small Cell Lung Cancer Tumor Mutational Burden Test Kit (NSCLC TMB Kit). In January 2020, the kit underwent the NMPA […]
price change in recent years
The selling price or list price of a medical device is driven by numerous factors, such as therapy area and complexity of procedure, level of reimbursement, development costs, the number of competing manufacturers, product life cycle, and whether there have been any recent new technology or product launches. In addition, the final selling pricing of […]
price change in recent years
The selling price or list price of a medical device is driven by numerous factors, such as therapy area and complexity of procedure, level of reimbursement, development costs, the number of competing manufacturers, product life cycle, and whether there have been any recent new technology or product launches. In addition, the final selling pricing of […]
Altaviz introduces new auto-injector platform for drug delivery
The auto-injector platform is intended for high-viscosity and high-volume biologics drug delivery. Credit: TShaCopy/Shutterstock.com. Medical device developer Altaviz has announced the launch of a new auto-injector platform, AltaVISC, for drug delivery. The new platform utilises Pico-Cylinders, developed by Altaviz’s affiliate company Picocy, to enable the delivery of both high-volume and high-viscosity therapies. AltaVISC can be […]
Heidelberg’s ANTERION platform gets US FDA approval
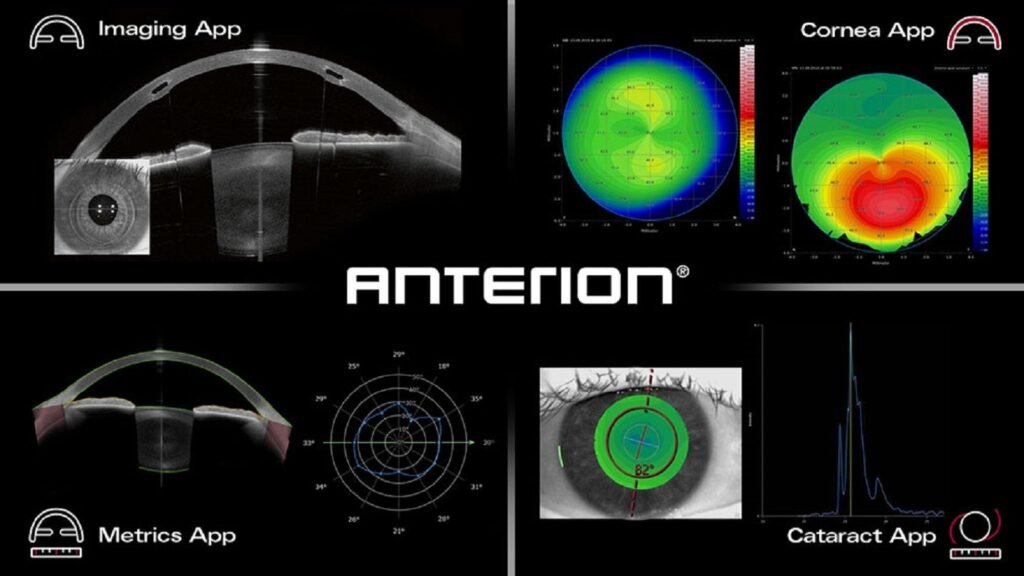
The ANTERION platform is intended to help streamline practice workflow. Credit: Heidelberg Engineering GmbH. Ophthalmic imaging company Heidelberg Engineering has secured US Food and Drug Administration (FDA) clearance for its ANTERION platform. ANTERION is an all-in-one upgradeable platform to transform anterior segment diagnostics and streamline practice workflow. It combines IOL power calculation, biometry with corneal […]
price change in recent years
The selling price or list price of a medical device is driven by numerous factors, such as therapy area and complexity of procedure, level of reimbursement, development costs, the number of competing manufacturers, product life cycle, and whether there have been any recent new technology or product launches. In addition, the final selling pricing of […]
