Philips woes worsen, FDA issues overheating safety warning for CPAP machine

The FDA decision follows the receipt of over 270 reports citing thermal issues such as fire, smoke, burns, and other signs of overheating while operating the machine. Image Credit: JHVEPhoto / Shutterstock. The US Food and Drug Administration (FDA) has issued a safety warning regarding the risk of overheating for the Philips Respironics’ DreamStation 2 […]
Unomedical’s VariSoft’s insulin infusion hit with FDA Class I recall tag
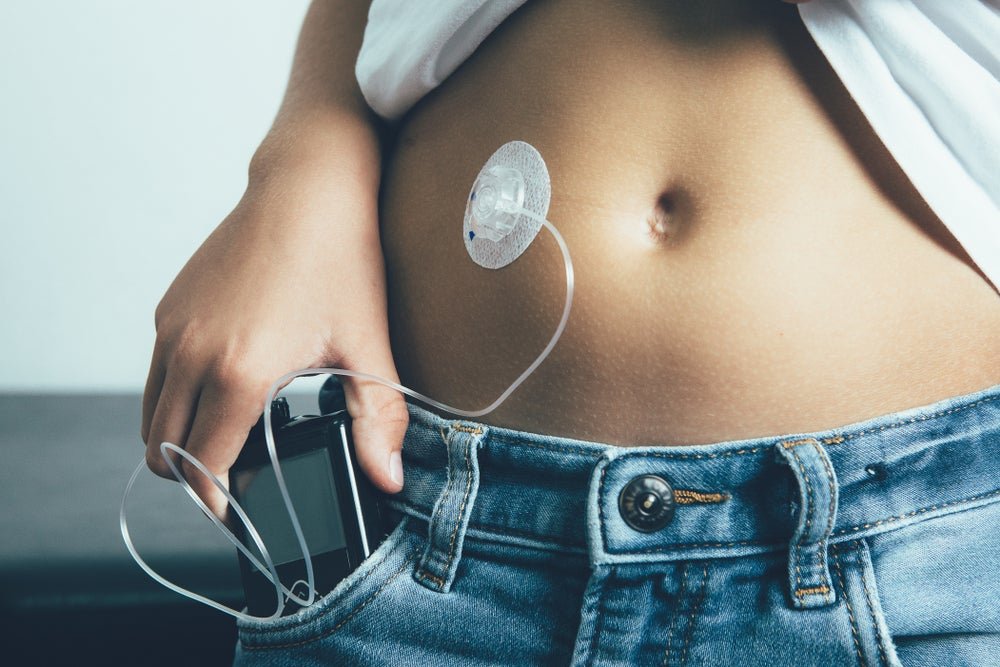
There have been high-profile cases of companies navigating diabetes device issues this year. Image credit: Shutterstock/Click and Photo. In a year that has seen a multitude of medical device recalls, Unomedical is the latest company to land a Class I tag after issues with its Varisoft infusion sets. The devices are used with insulin pumps […]
Phillips woes worsen, FDA issues overheating safety warning for CPAP machine

The FDA decision follows the receipt of over 270 reports citing thermal issues such as fire, smoke, burns, and other signs of overheating while operating the machine. Image Credit: JHVEPhoto / Shutterstock. The US Food and Drug Administration (FDA) has issued a safety warning regarding the risk of overheating for the Philips Respironics’ DreamStation 2 […]
First surgeries testing rotator cuff augmentation implant carried out in Florida
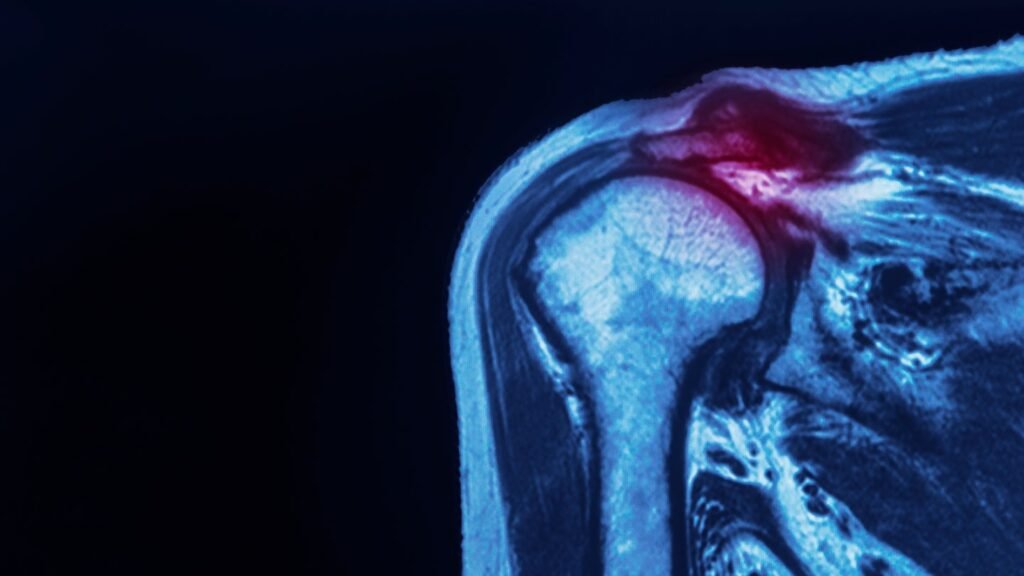
The announcement follows after the implant received 510(k) clearance from the US Food and Drug Administration in August of this year. Credit: Shutterstock / Peter Porrini The first surgeries testing an implant system designed to protect injured tendons have been successfully carried out at a Florida hospital ahead of the implants US rollout. Anika Therapeutics’ […]
Philips launches new MR Smart Fit coils at RSNA
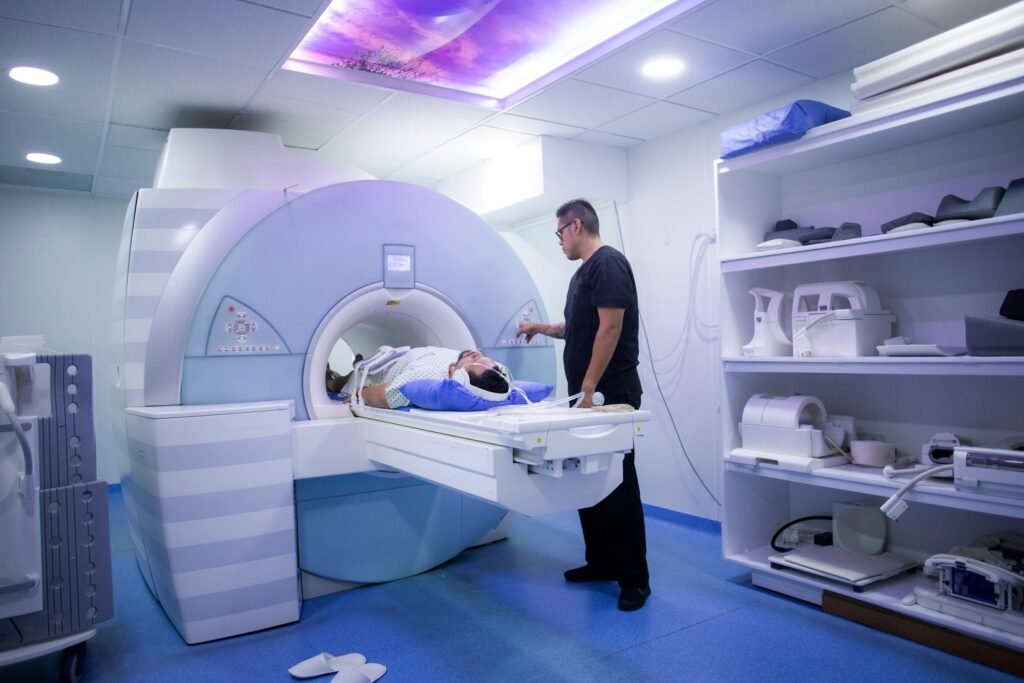
MRI RF coils are essential components for every MRI examination. Credit: DarioGaona via Getty Images. Royal Philips has introduced three new magnetic resonance (MR) Smart Fit coils at the Radiological Society of North America (RSNA), in Chicago, US. The new solutions include the Smart Fit Torso Cardiac 1.5T coil, the Smart Fit 1.5T shoulder coil, […]
Spinal Elements introduces Ventana 3D-Printed Interbody portfolio

Ventana products come with a uniquely designed implant architecture. Credit: Spinal Elements. Spine technology company Spinal Elements has commercially introduced its Ventana 3D-Printed Interbody portfolio. The portfolio, which has been launched completely, consists of the Ventana P/T Posterior Lumbar Interbody System, the Ventana L Lateral Lumbar Interbody System, and the Ventana C Anterior Cervical Interbody […]
Clario introduces AI-powered tool for cardiac safety evaluations in trials
The approach has been tested on more than 80,000 Phase I volunteers. Credit: Gerain0812 via Shutterstock. Clario has introduced the ECG Quality Score tool, powered by artificial intelligence (AI), for improving cardiac safety evaluations in clinical trials. The new tool includes the Early Precision QT Quality Summary Report and the Early Precision QT Quality Detailed […]
Pharma’s path to Net Zero: Targeting Scope 3 emissions

Pharmaceutical companies need to address Scope 3 emissions to follow the path to net zero. Credit: sarawin aexmo via www.shutterstock.com. Net zero emission targets have never been more relevant to society. This year alone, there have been 24 extreme weather events in the US that have cost at least $1bn, as per the US National […]
Inflammatix receives Breakthrough Device Designation for TriVerity system

The system includes a panel of 29 messenger RNAs. Credit: Inflammatix. Inflammatix has received breakthrough device designation from the US Food and Drug Administration (FDA) for its TriVerity acute infection and sepsis test system. Featuring the Myrna instrument and the TriVerity test, the new system is presently under development for use in emergency departments to […]
Penumbra enrols first patient in trial of Lightning Flash
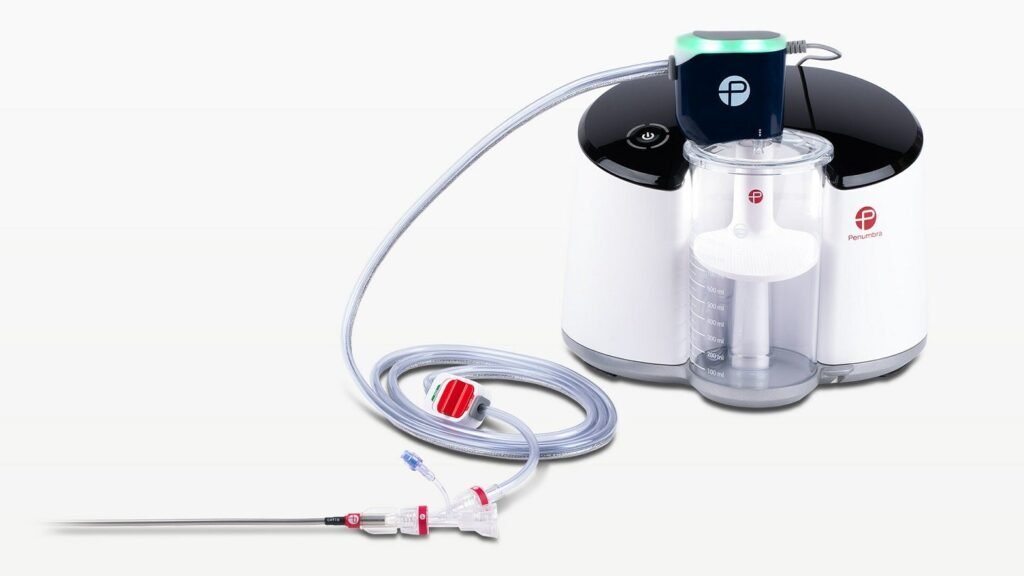
Approximately 900,000 cases of symptomatic PE occur each year in the US. Credit: Penumbra/PRNewswire. Penumbra has enrolled the first patient in the STORM-PE study, which is designed to assess anticoagulation alone against anticoagulation plus Lightning Flash to treat pulmonary embolism (PE). In collaboration with the PERT Consortium, the multicentre, prospective, and randomised controlled trial intends […]
