Signal: FTC granted injunction to pause IQVIA’s acquisition of Propel Media

IQVIA’s acquisiton of PMI is unlikely to go ahead following the FTC’s injunction. Photo: OleksSH/Shutterstock. The US Federal Trade Commission (FTC) has been granted a temporary injunction against health tech giant IQVIA’s acquisition of digital advertising company Propel Media and its healthcare marketing subsidiary DeepIntent due to fears it may lead to anti-competitive practices. IQVIA, […]
LumiThera requests FDA for de novo reclassification of ocular device

Valeda is already available in Europe, having received the CE marking in 2018. Image credit: Shutterstock/fizkes. US-based LumiThera has submitted a de novo request to reclassify the Valeda light delivery system as a Class II device, following the feedback from the FDA. Under the request process, the system can be assigned an official classification as […]
GlucoTrack eyes human trials for implantable blood glucose monitor

GlucoTrack aims to develop a continuous glucose monitor device with a long sensor life, no additional wearable component, and a once-only calibration event. Image Credit: Buravleva stock / Shutterstock. New Jersey-based GlucoTrack has completed the first preclinical study with its implantable continuous glucose monitor (CBGM) and is preparing for first-in-human studies, with trial initiation expected […]
FDA approves AnX Robotica’s AI endoscopy tool

The AI-based tool identified abnormalities at a higher sensitivity compared to conventional readings by gastroenterologists. Credit: Pikovit44 via Getty Images. US-based manufacturing company AnX Robotica has received de novo clearance from the US Food and Drug Administration (FDA) for its artificial intelligence (AI)-assisted endoscopy tool NaviCam ProScan. NaviCam Proscan utilises a deep learning algorithm called […]
Daxor seeks US FDA approval for blood volume analyser
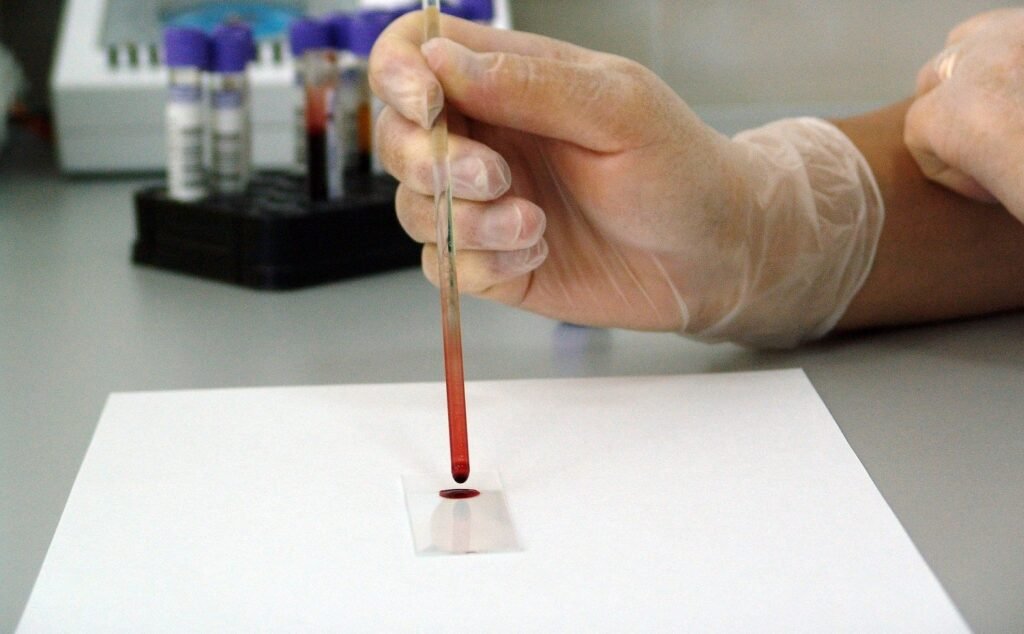
The BVA-100 provides safe, accurate, objective quantification of blood volume status. Credit: PublicDomainPictures from Pixabay. Daxor Corporation is seeking approval from the US Food and Drug Administration (FDA) for its next-generation blood volume analyser, Daxor BVA. Daxor BVA is designed for rapid, simple, and bedside patient care. The company submitted to the FDA through the […]
CGBio gets breakthrough designation from US FDA for NOVOSIS PUTTY

The designation acknowledges potential of NOVOSIS PUTTY to offer more effective treatment for spine implantation. Credit: Gundula Vogel from Pixabay. South Korean bio-regenerative medicine company CGBio has received the US Food and Drug Administration’s (FDA) breakthrough device designation for its NOVOSIS PUTTY, a bone substitute material. NOVOSIS PUTTY is the first Korean-developed implantable device for spine […]
CardioMech secures funding for chordal repair technology

CardioMech’s device could potentially reduce the need for open-heart surgery. Credit: StockSnap from Pixabay. Norway-based medical device company CardioMech has secured $13m in a funding round to advance its transcatheter mitral valve chordal repair technology. The heavily oversubscribed funding round attracted both existing and new investors, including an undisclosed strategic investor. This brings the total […]
Angle secures Eisai contract for Portrait HER2 assay

The pilot study’s success could lead to multiple large-scale follow-up studies. Credit: maleni ferrari from Pixabay. Liquid biopsy company Angle and global pharmaceutical company Eisai have signed a contract for a pilot study involving the former’s Portrait human epidermal growth factor receptor 2 (HER2) assay. The study will be part of a Phase II clinical […]
EndoSound secures FDA clearance for endoscopic ultrasound technology

The device received FDA breakthrough device designation in July 2021. Image credit: Shutterstock/Peter Porrini. The US Food and Drug Administration (FDA) has granted 510(k) clearance to EndoSound’s Vision system, an endoscopic ultrasound device. Endoscopic ultrasounds are minimally invasive procedures that use ultrasound to assess the upper gastrointestinal tract. High-frequency sound waves produce detailed images of […]
Probo Medical to acquire Alpha Source Group

The acquisition will enable Probo to broaden its diagnostic imaging solutions platform. Credit: HealthyCapture Studio/Shutterstock.com. Diagnostic imaging equipment distributor Probo Medical has entered a definitive agreement to acquire Alpha Source Group, a services provider for the North American healthcare market. With the acquisition, Probo will be able to significantly broaden its diagnostic imaging solutions platform. […]
