Venture funding down by 31% compared to last year

VC funding in 2023 is down by 57.4% from its peak of a total of $49.3bn in VC financing deals in 2021. Image Credit: iSOMBOON / Shutterstock. After a successful 2021 and a muted 2022, the past year saw a further decline in venture capital (VC) financing in the medical device space. Several factors led […]
Siemens secures FDA approval for new MRI scanner
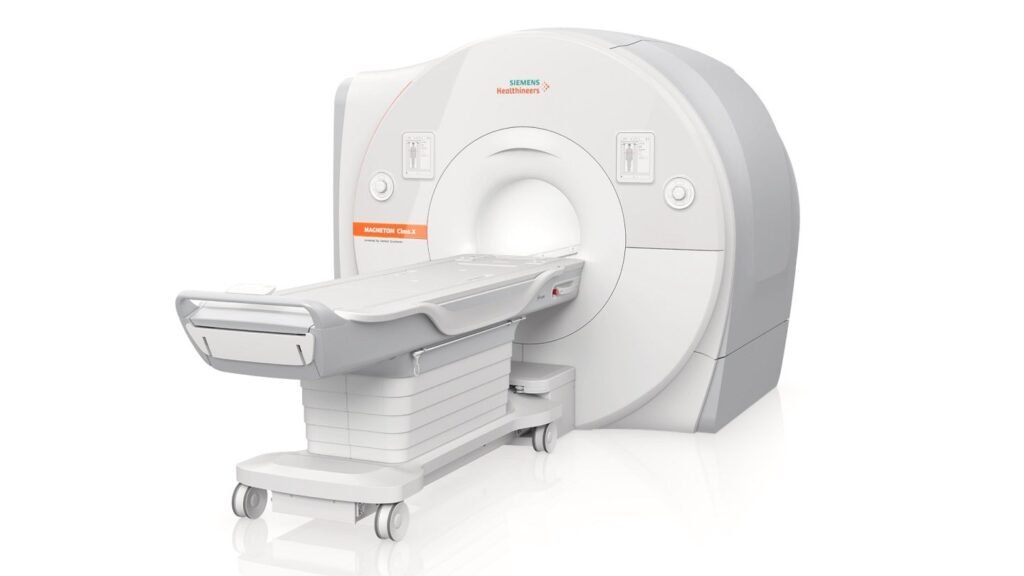
Some of the features of the MRI scanner device include the Deep Resolve reconstruction technology and myExam Companion workflow solution. Credit: Siemens Medical Solutions USA. Siemens Healthineers has received approval from the US Food and Drug Administration (FDA) for its MAGNETOM Cima.X 3 Tesla (3T) magnetic resonance imaging (MRI) whole-body scanner. The device features what […]
Pylo Health introduces new devices for remote patient monitoring

The company’s new Pylo 900-LTE blood pressure monitor and 200-LTE weight scale. Credit: Pylo Health/ PRNewswire. Pylo Health has launched two cellular-connected remote patient monitoring devices, the Pylo 900-LTE blood pressure monitor and the Pylo 200-LTE weight scale. The two devices are validated clinically and designed to integrate with the Prevounce care management platform and […]
SpineX’s neuromodulation device shows improvement in adult cerebral palsy
SpineX received breakthrough device designation from the FDA for the SCiP device for children with cerebral palsy in May 2022. Image credit: Shutterstock/Yurchanka Siarhei. SpineX has announced positive results from a first-in-human study evaluating the use of its electrical neuromodulation device in a patient with cerebral palsy. The results, which were published in BioElectronic Medicine, […]
Geneseeq scores FDA breakthrough designation for cancer detection device
Geneseeq’s CanScan test can be used to detect a variety of cancer types and can obviate the need for multiple tests. Image Credit: Connect world / Shutterstock. The US Food and Drug Administration (FDA) has granted breakthrough device designation to CanScan, the multi-cancer early detection solution by Canadian medtech Geneseeq. CanScan was built using Geneseeq’s […]
SEKISUI signs Covid-19 test distribution deal with Aptitude in US

The Metrix Covid-19 Test is designed for the qualitative detection of nucleic acid from the SARS-CoV-2 virus. Credit: SEKISUI Diagnostics. SEKISUI Diagnostics has signed an exclusive distribution agreement with decentralised molecular diagnostics developer Aptitude Medical Systems to sell the Metrix Covid-19 test in the US market. Said to be the only Emergency Use Authorization (EUA)-approved […]
FDA grants breakthrough device designation for Renovos’ bone graft gel
The US bone grafts and substitutes market is forecast to grow to $2.18bn in 2030. Credit: Shannon Fagan via Getty Images. UK-based Renovos Biologics has been granted breakthrough device designation from the US Food and Drug Administration (FDA) for Renovite BMP-2 (bone morphogenic protein 2), a synthetic nanoclay bone fusion gel. Renovite BMP-2 is used […]
Enspectra’s VIO System secures FDA 510(k) clearance

The device is designed for use by dermatologists or pathologists trained in skin histology. Credit: backy3723 from Pixabay. Enspectra Health has received FDA 510(k) clearance from the US Food and Drug Administration (FDA) for VIO System, a non-invasive skin imaging technology. By combining reflectance confocal and multiphoton laser scanning microscopy, the VIO System can produce […]
Weill Cornell Medicine secures grant for breast cancer blood test

Weill Cornell Medicine’s breast medical oncology director Dr Massimo Cristofanilli is the study’s principal investigator. Credit: Weill Cornell Medicine. Researchers at Weill Cornell Medicine have received a $2.4m grant to validate a blood test intended for early breast cancer detection. The grant from the US Department of Defense Breast Cancer Research Program will support the […]
Nalu raises $65m funding for neurostimulation system expansion
The company focuses on providing a minimally invasive and non-opioid treatment option for individuals with chronic neuropathic pain. Credit: Engin Akyurt from Pixabay. US-based Nalu Medical has raised $65m in a Series E financing round to further the development and commercialisation of its neurostimulation system for chronic pain treatment. The funding will primarily accelerate commercial […]
